The Essure birth control device has received an unusual number of patient complaints since its approval in 2002, finally prompting the FDA in September 2015 to convene an advisory panel to examine the safety issues with the device.
Essure is the first permanent birth control device designed to be placed through a minimally invasive procedure. The procedure can be done in a doctor’s office: the device is implanted via a catheter through the vagina, cervix and uterus, into the Fallopian tubes. Once there, it induces inflammation that causes tissue growth that blocks the Fallopian tubes and prevents sperm from entering the tubes and getting to the eggs. Essure is made out of plastic resin (polyester), stainless steel, and nickel-titanium alloy.
The FDA has received over 5000 complaints of adverse events with the Essure device, and this is likely only the tip of the iceberg, since many adverse events never get reported to the FDA. A Facebook group formed by women who experienced severe problems after having Essure implanted has over 22,000 members.
Problems with Essure
The problems reported with Essure range from mild to severe. Women have experienced severe chronic pelvic pain, heavy bleeding, pain with intercourse, extreme bloating, digestive complaints, depression, hair loss, fatigue, allergic reactions to nickel, and autoimmune problems. In some women, the device perforates the Fallopian tubes and ends up in lodged in different areas of the abdomen or uterus, and in some cases, hysterectomy is required to treat the complications produced by Essure. A recent study has shown that women who have Essure implanted are 10 times as likely to require followup surgery within the next year, including major surgeries like hysterectomy, compared to women who have tubal ligation performed laparoscopically. In addition, the company selling Essure says the device is over 99 percent effective at preventing pregnancy, but some studies have shown that it may only be about 90 percent effective.
Many women who have experienced problems with Essure are calling for the device to be removed from the market. However, some doctors say that Essure is an important, safe and effective birth control option for women. Whether this is true or not should have been well addressed with clinical trials before the device was approved by the FDA. However, this device was approved under the FDA’s “fast track” review process, and perhaps the device was approved too quickly and with too little information about the long term safety and effectiveness.
Was Essure Approved Too Quickly?
The fast track review process was designed to allow the FDA to expedite reviews for drugs or devices that are “first in class products” that provide noticeable clinical advances. Essure qualified for this process, since it is the first permanent sterilization device that can be implanted with a minimally invasive procedure done in a doctor’s office. And indeed, the concept seems like a great advance for women who desire permanent, effective birth control without surgery—the only other permanent sterilization methods for tubal ligation require a general anaesthetic and a laparoscopic surgery.
However, some doctors have argued that the FDA’s fast track review process has allowed the FDA to approve drugs and devices that have proved harmful, or ineffective, or both. For example, from 1992 to 2010, the FDA approved 35 new oncology products, for 47 different clinical indications, using an expedited review process. Part of the expedited review process relies on companies continuing to do clinical trials and surveillance post-approval. In the post-approval trials, only 26 of the 47 indications were confirmed to have clinical benefit, and confirmatory trials were never even performed for 14 of the indications.
Moving Forward–Are the Proposed Steps Enough?
With Essure, the FDA advisory panel has suggested some actions that may help mitigate the potential risks associated with it. For example, a patient registry was suggested, in order to track the rates of adverse events and pregnancies. It was also suggested that training programs for device implantation and removal be implemented. Currently, many doctors are not trained in removal of the device, making it even more difficult for patients who have problems with it. The panel stated that a checklist or signed informed consent form should be required, to ensure that patients are adequately informed of the risks, benefits, and alternatives. And guidelines should be developed, to assist doctors in identifying problems earlier. The FDA is not required to implement the suggestions of its advisory panels, but it often does.
There is no doubt that safe and effective birth control options are very important for women’s health, but I question whether most women would have valued a faster approval process for Essure over having it well tested for safety and effectiveness. It seems reasonable to wonder whether a device made out of plastic and highly allergenic metal, that is designed to induce inflammation in the body, implanted there permanently, should have undergone more complete testing before being introduced to the market. And even if the FDA does implement the above actions, is that enough to protect women who may be harmed by this device?




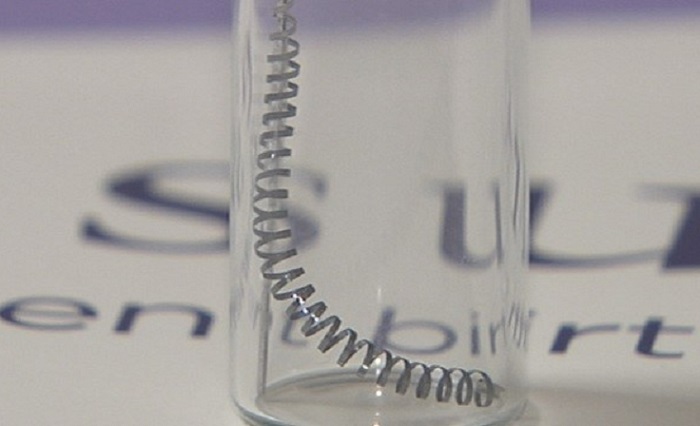








I has my essure implants inserted 10 years ago. last year one of the implant fell out. I want to know if its safe to have only 1 in. and should I have the other one remove. I do want another baby
You will need to discuss that with a doctor. The implants are designed to create scar tissue that will close your Fallopian tubes. So even with one implant out, both tubes are likely still closed.