Some individuals experience negative reactions and the worsening of symptoms when they begin thiamine repletion therapies using the more biologically available formulations like thiamine tetrahydrofurfuryl disulfide (TTFD). Dr. Lonsdale calls these paradoxical reactions. In this article, I examine the chemistry beyond these reactions and provide some hypotheses regarding why they happen and how to mitigate them.
TTFD Basics
In order to understand why some individuals react negatively to TTFD supplementation, it is essential to understand the basics behind TTFDs molecular configuration and how it is processed by cells. The primary difference between ordinary thiamine and TTFD is an extra chemical group called a mercaptan group. The mercaptan is derived from allicin, a compound found in garlic, and is connected to the thiamine molecule via a special sulfur-sulfur bond called a disulfide bond. Importantly, it is this unique chemical group that accounts for TTFD’s ability to traverse membranes in the body without the need for a transport system.
Upon ingestion, TTFD is mostly absorbed into the blood from the gastrointestinal tract in whole form as TTFD. As it travels through the blood, it can penetrate the brain and other organs without cellular transporters. One of the main sites of absorption is the red blood cells. Upon penetration of the red blood cell membrane, TTFD must first be processed or “broken apart” before it can release the thiamine contained within its chemical structure. After thiamine is released into the cell, the ancillary mercaptan group must also be processed and/or detoxified through alternative pathways. It is therefore theoretically plausible that errors involved in the processing of TTFD could contribute toward negative side effects or reactions to this nutrient.
How TTFD Is Processed Inside the Cell: The Glutathione Connection
Of the few molecules which have been shown to reduce TTFD, glutathione performs this function most effectively. As the cell’s primary antioxidant, glutathione is responsible for donating electrons to neutralize reactive oxygen species, and can either be found in its reduced form or its oxidized form. Once a reduced glutathione molecule (GSH) has donated its electron, it bridges with another to molecule to form oxidized glutathione (GSSG). GSSG is then recycled back to two GSH molecules through accepting electrons from NADPH via the enzyme glutathione reductase (vitamin B2 as FAD dependent). 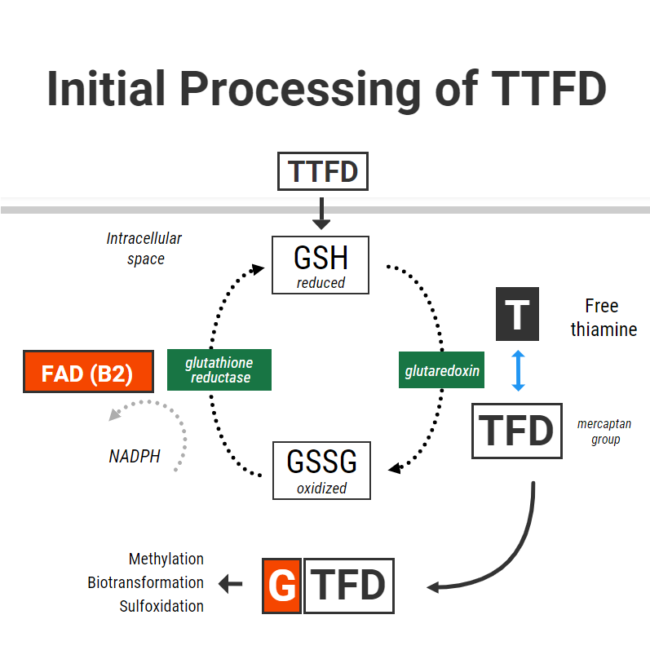
When TTFD enters cells, GSH in red blood cells chemically reduces TTFD via a process called “disulfide exchange” (presumably using a protein called glutaredoxin). Reduced glutathione becomes oxidized glutathione and TTFD “releases” thiamine to producing free thiamine inside the cell with an extra TFD mercaptan group left over.
The initial phase of processing TTFD requires that cells have enough reduced glutathione. Furthermore, the more GSH you have – the faster the rate of this reaction. So in simple terms, to obtain thiamine from TTFD the cells “use up” their reduced glutathione.
I recently had correspondence with one individual who only gained tolerance of TTFD after supplementing with 200mcg of selenium in the form of sodium selenite. Selenium supplementation in different forms has been shown to increase red blood cell GSH levels by up to 35%. This is thought to occur due to selenium’s ability increase glutathione synthesis through upregulating the enzyme gamma-glutamylcysteine synthetase. I suspect that poor glutathione status might be one of the reasons for benefit from selenium.
Having enough glutathione is clearly very important, but recycling it is also essential to maintain a pool of glutathione in its reduced form. Unfortunately TTFD can place a burden on this system, and this was demonstrated in one old study from Japan which showed that TTFD administration rapidly lowered red blood cell GSH. Interestingly enough, that same experiment showed that GSH levels were restored within 5-10 minutes. This restoration was accomplished by the vitamin B2 (as FAD)-dependent enzyme glutathione reductase, which donates electrons to GSSG with the reducing power of NADPH to recycle it back to two GSH.
What this basically means is that cells require a robust antioxidant system to properly process TTFD and return back to their original state. First, cells need enough of the antioxidant GSH to cleave thiamine. Second, cells also need to be able to recycle the oxidized glutathione back to its reduced state.
Poor Glutathione Status and Difficulty With TTFD
In someone who has poor glutathione (GSH) status, they might theoretically be less able to cleave thiamine from TTFD. There are many reasons why someone may have poor glutathione status:
- Low precursors (cysteine, glutamate, glycine)
- Chronic oxidative burden and/or inflammation
- Deficiencies in the nutrients required to generate, process, or utilize glutathione (B6 or selenium)
A total and/or functional riboflavin deficiency is the probably the most common culprit responsible for poor glutathione reductase activity. The glutathione reductase enzyme also requires adequate reducing power from NADPH to drive the enzymatic reaction. NADPH is derived from niacin (vitamin B3) and is generated in the pentose phosphate pathway which, ironically, requires the thiamine-dependent enzyme transketolase.
In the context of poor enzyme activity, without the reducing powder to drive GSSG back to GSH, the oxidized form of glutathione can theoretically drift towards the path of generating a free radical called the glutathione radical. This alone could further contributes to oxidative stress and cell damage.
Below is a hypothetical scenario to demonstrate my point:
- An individual suffers from long-term thiamine deficiency and has suboptimal riboflavin status
- Thiamine deficiency leads to lower activity of transketolase
- Low transketolase activity produces a lack of NADPH
- A lack of NADPH and a lack of FAD means that glutathione reductase is unable to efficiently recycle glutathione, which produces an imbalance between reduced/oxidized glutathione.
- Intracellular GSH is further lowered by taking high dose TTFD, and there is not enough enzyme activity to recycle it back
- Oxidative stress is made worse
In the above scenario, taking a high dose of TTFD may not be appropriate. Rather, restoring NADPH levels through supplementing with ordinary thiamine and supporting the glutathione system via other measures might be advised before starting with TTFD. Optimal riboflavin status is also necessary for the above processes to run smoothly.
Older research in Japan showed that TTFD supplementation could lead to a secondary B2 deficiency through increased urinary excretion. The increased need for glutathione reductase could at least also contribute to this effect. When taking TTFD, it has downstream effects on other nutrients. Hence, these supporting nutrients should also be taken in conjunction when someone is supplementing TTFD in high doses.
Some basic laboratory measurements of glutathione status include:
- Whole blood glutathione (low)
- Gamma-glutamyl-transpeptidase (high)
- Urinary pyroglutamic acid (high)
Furthermore, there are several functional markers which can be measured to assess riboflavin status, including direct measurement of red blood cell glutathione reductase activity:
- Urinary glutaric acid (high)
- Whole blood B2
- Urinary adipic, suberic, ethylmalonic acids (high)
- Urinary succinic acid (high) can also be suggestive along with a few other organic acids
- Erythrocyte glutathione reductase activity (low)
To summarize, the initial cellular processing of TTFD requires adequate levels of reduced glutathione. Glutathione becomes oxidized, and so TTFD has can have a depleting effect on GSH and increase the requirement for recycling. If there is insufficient active B2 (as FAD) or NADPH levels, glutathione is not likely to be recycled sufficiently and may lead to GSSG radical formation.
It is therefore possible that the glutathione-depleting effect of TTFD could be responsible for some of the side effects associated with supplementation. This is probably most applicable in individuals with poor glutathione recycling and underlying oxidative stress. Therefore, nutrient therapies that may support this initial phase of TTFD metabolism include:
- Selenium (improve GSH levels)
- Riboflavin (improve GSSG-GSH recycling)
- Niacin (increase NADPH)
- Ordinary thiamine (increase NAPH via PPP)
- NAC, glycine and/or glutathione TAKEN HOURS AWAY from TTFD (GSH precursors)
We Need Your Help
More people than ever are reading Hormones Matter, a testament to the need for independent voices in health and medicine. We are not funded and accept limited advertising. Unlike many health sites, we don’t force you to purchase a subscription. We believe health information should be open to all. If you read Hormones Matter, like it, please help support it. Contribute now.