I was recently invited to present my research on the HPV vaccine at the Euroscicon Controlling Cancer Summit held in London on 12 May 2014. The theme of the presentations was Advances in Cancer Screening and Prevention Research and the paper I presented was titled: HPV vaccines have not been demonstrated to be safe and effective in the prevention of cervical cancer.
HPV Infections Mostly Harmless
In my presentation I provided evidence that HPV infections are harmless and asymptomatic unless specific environmental co-factors are also present. This is why HPV infections should not be feared by the public and why the medical literature states that cancer is a rare outcome from any type of HPV infection. The fact that cervical cancer is a higher risk in developing countries than in developed countries is explained by the presence of environmental co-factors that are necessary for an HPV infection to progress to cancer. These co-factors (risk factors) are more prevalent in the developing countries. The fact that HPV infections are mostly harmless on their own means that vaccinating all women in developed countries (e.g. Australia, US and the UK) results in the majority of women (99%) being on a drug for a disease that they are not at risk of getting. This is not cost-effective and it is also not necessary because the vaccine has not been proven to be safer or more effective than Pap screening combined with surgery.
If the HPV vaccines are proven to have value in years to come it could be offered to women in the high-risk category. That is, women who are exposed to the environmental co-factors that are necessary for an HPV infection to progress to cervical cancer. However, as yet the vaccine has not been proven to be safe or effective in preventing cervical cancer. Currently governments are claiming that because the HPV vaccine targets 2 of the 15+ strains of HPV associated with causing most cervical cancer, it will prevent some cervical cancer, but they have not determined how much can be prevented. This argument is flawed if the majority of women on the drug are not at risk of cervical cancer and if there is already an effective method of preventing cervical cancer in place. In this case, Pap screening combined with surgery is an effective method of prevention (9 out of 10 cancers) and it is risk free and will still be required by vaccinated women.
The Global Harm Associated with HPV Vaccines
Currently there is much global debate about the harm that is being associated with HPV vaccination programs. As of June 2014 Japan has stopped recommending this vaccine until further safety studies have been conducted. India and Utah have also stopped recommending this vaccine and France is considering similar action. In France the use of HPV vaccine was debated in an open scientific forum on 22 May 2014. This forum allowed all stakeholders to present their case to the French parliament. This is the debate that governments and health professionals are not having in many other countries, for example, Australia. In fact, the Australian government is recommending this vaccine free to all adolescent girls and boys in school programs without a debate about its safety and efficacy in preventing cervical cancer (a non-infectious disease).
HPV Vaccine Adjuvants
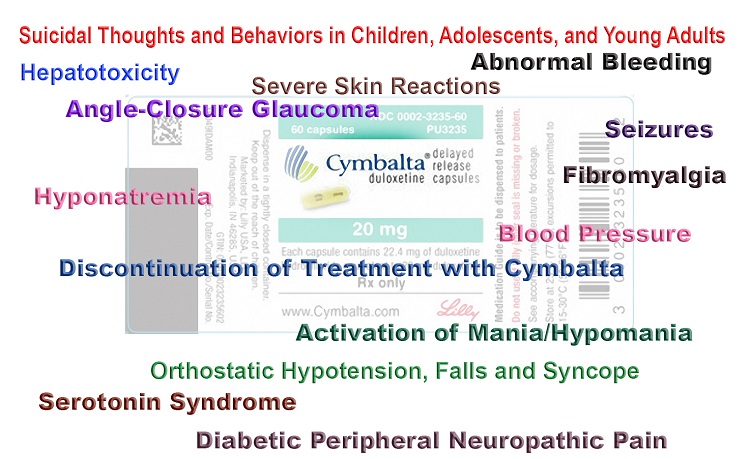
The HPV vaccine has two ingredients that are linked to causing infertility. These are sodium borate and polysorbate 80 and the Australian government has not explained why these ingredients are in a vaccine that is being recommended free to adolescents. This vaccine also has three times as much aluminium hydroxyphosphate sulphate (an adjuvant that is linked to autoimmune diseases and hypersensitivity) as any other vaccine and three times as many adverse events have been reported to this vaccine. The most common adverse events are neurological conditions and autoimmune diseases.
Adverse Events Associated with HPV Vaccines
Since the introduction of HPV vaccines 34,700 adverse events have been voluntarily reported to the US CDC, including 157 deaths and 6,977 permanent disabilities and chronic illness. This is possible because the Merck (vaccine manufacturer) funded Phase 3 clinical trials for Gardasil vaccine did not use an ‘inert’ (non-active) placebo in the unvaccinated control group. They used aluminium adjuvant in the comparison group and they did not collect long-term adverse events. The clinical trials only followed the health outcomes of all vaccinated girls actively for 15 days after vaccination. After this time the reporting of AE’s was voluntary which does not allow scientists to make causal relationships to the vaccine.
Here is a link to a video of the serious adverse events that some girls have experienced after using this vaccine. These have included seizures, paralysis, convulsions, tics, encephalopathy, chronic fatigue syndrome and death. The parents of injured children and those that have died after vaccination urge you to research this vaccine before you trust the government’s recommendation of this vaccine.
Report from the French Parliament on the Safety of Aluminium Adjuvant in HPV Vaccines (22 May 2014)
The public hearing held in Paris on the safety of aluminium adjuvants in vaccines was attended by the French Health Minister and reported on by the European parliament. The hearing was open to the press and titled ‘Vaccine Adjuvants: A Controversial Question’. The most recent science on aluminium adjuvants in vaccines demonstrates that many individuals have a pre-disposition (genetic condition) to experiencing a serious reaction from aluminium adjuvants in vaccines. These serious reactions include neurological damage and autoimmune diseases – multiple sclerosis, arthritis, lupus, etc – and are caused by the artificial stimulation of the immune system with vaccines. Here is a link to the report on the public hearing http://sanevax.org/french-vaccine-debates-immediate-measures-required/
This indicates the significance of fully informing parents about the vaccines that are recommended in government vaccination programs and the importance of vaccines being administered by general practitioners with an assessment of the family history of the patient. Vaccines are a medical intervention and they should not be administered in school programs because family history is a contraindication to vaccination.
Conclusion
Cervical cancer is curable with early detection by Pap screening (9 out of 10 cancers) and all vaccinated women will still need Pap screening. This is because the vaccine (costing $Au450 per person) does not target ~30% of cervical cancer (13+ strains of high-risk HPV are not covered in the vaccine) – even if it is proven to be of some value in years to come. It is also a fact that HPV infections are harmless unless specific environmental co-factors are also present and this is why vaccinating all women in developed countries results in the majority of women (99%) being on a drug for a disease they are not at risk of getting.
Reference
Wilyman J, 2013, HPV vaccination programs have not been shown to be cost-effective in countries with comprehensive Pap screening and surgery. Infectious Agents and Cancer. 8:21 (June): pp1-8.
About the Author: Judy Wilyman MSc, is a PhD Candidate studying Population Health Policy at the University of Wollongong (UOW) School of Social Sciences, Wollongong, Australia. She is the founder of Vaccination Decisions, a website that she has set up to present her research.
Participate in Research
Hormones MatterTM is conducting research on the side effects and adverse events associated with Gardasil and its counterpart Cervarix. If you or your daughter has had either HPV vaccine, please take this important survey. The Gardasil Cervarix HPV Vaccine Survey.
To take one of our other Real Women. Real Data.TM surveys, click here.
To sign up for our newsletter and receive weekly updates on the latest research news, click here.
What Else Can I Do To Help?
Hormones MatterTM is completely unfunded at this juncture and we rely entirely on crowdsourcing and volunteers to conduct the research and produce quality health education materials for the public. If you’d like help us improve healthcare with better data, get involved. Become an advocate, spread the word about our site, our research and our mission. Suggest a study. Share a study. Join our team. Write for us. Partner with us. Help us grow. For more information contact us at: info@hormonesmatter.com.
To support Hormones Matter and our research projects – Crowdfund Us.








Thank you very much for providing clear & concise information do parents of girls like myself can make informed decisions.