Some 20 years ago, during my very first neuro class taught by an accomplished neurologist from a prominent research university, I had a conversation about hormones and the brain. It was a brief conversation during which he admitted not only knowing nothing about how hormones affected the brain or nervous system functioning, but also, how he and others had no interest in considering the question. He believed hormones were too complicated to consider relevant. One didn’t ‘mess with hormones’ as he put it.
Lucky for us, some intrepid neurologists have moved the science of neuroendocrinology past the foibles of ‘don’t mess with hormones’ to hormones might be important therapeutic options. Nowhere is this more evident than in the areas of traumatic brain injury and diseases of demyelination. Here we see advances in hormones used as viable and important treatments where once there were none. Although the research is yet in its infancy and suffers from the typical one-size-fits-all approach, it marks a huge step forward in clinical neuroendocrinology.
What is Neuropathic Pain?
Neuropathic pain, the often chronic and difficult to treat pain that comes from nerve injury and demyelination affects approximately 3% of the population. The number of individuals suffering from neuropathy is likely much higher when one considers diseases such as endometriosis and the ill-understood, under-recognized neuropathy emerging post medication or vaccine adverse reactions. The experience of neuropathic pain in hands, feet, arms and legs is described as burning, freezing, electrical, tingling, prickling and more often than not, severe and unrelenting. As the nerve injury progresses and the pain continues, the rawness and intensity of the pain becomes indescribable to someone who has not experienced nerve pain firsthand.
Hormones and the Nervous System
Since the late eighties, researchers have known that steroid hormones, such as progesterone were not limited to reproductive functions and that many steroids were active in the nervous system. Not only were those steroids synthesized peripherally in ovaries, adrenals or adipose tissue able to cross the blood brain barrier, but all the core substrates for steroid synthesis were available in the brain too, meaning the brain could make its own steroids, de novo, from scratch. Researchers initially deemed steroids made or active in the brain as neurosteroids. Eventually, that nomenclature fell by the wayside as researchers realized there was tremendous crosstalk between peripheral and central hormones, no matter where the hormones were synthesized.
It should be noted, that hormones exert influence all over the body and brain via receptor binding. (A discussion on hormones and receptors can be found here: Promiscuous Hormones and Other Fun Facts.) In addition to steroid hormone receptors on (cell membrane) and in (nuclear) hormone-specific cells, like those in ovary, testes, adrenals, uterus, endometrium, hormone receptors are co-located on neurons, glial cells, oligodendrocytes and Schwann cells (myelin producing cells), immune cells, cardiomyocytes (heart), hepatocytes (liver), adipocytes – essentially every cell, organ or tissue in our body is modulated in some way by a hormone. Hormone influence is particularly important in the in the nervous system, where everything from neurotransmitter release and uptake to synaptic connections are modulated.
Traumatic Brain Injury, Peripheral Neuropathy and Hormones
When we talk about injuries to the nervous system, be it the brain and spinal cord, which is called the central nervous system (CNS), or all of the nerves that control movement and organ function in the body, the peripheral nervous system (PNS), there are two categories of injuries, those that develop acutely, post trauma, or those that develop chronically because of some metabolic dysfunction. In the case of the former, traumatic brain injuries (TBI) or traumatic nerve injuries, the research points to progesterone for repair and regrowth. In the case of the later, where injuries develop as a result of internal and often chronic dysfunction, such as diabetic neuropathy, multiple sclerosis and other diseases affecting nerve fibers and myelin, less is known about progesterone and the thyroid hormone triiodothyronine (T3) is implicated, more strongly.
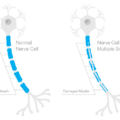
What is Myelin and How Does it Impact Neuropathy?
Myelin is the insulation that protects the axons of the neuron (in the brain) or nerve (in the body) to allow rapid conduction or messaging across the brain or through the body. Recall the axon is the part of the neuron/nerve that sends messages to other neurons/nerves, to other tissues, like muscle, or to organs like the heart and the liver. The dendrites receive messages and the nucleus processes messages. Myelin is like the plastic coating around the electrical wiring in your house. If the coating is too thick, conduction is blocked. If the coating is frayed or too thin, electrical sparks fly everywhere. Frayed myelin around axons is one of the mechanisms of neuropathic pain. Myelinated axons in the brain look white and therefore are called white matter. Whereas the grey matter, is where the nuclei of the brain reside. White matter in the brain consists of the oligodendrocytes – the type of cell that forms the myelin sheathing around axons. Myelin in the body, around the peripheral nerves, is made from cells called Schwann cells.
Progesterone, Myelination and the Nervous System
In the 1990s, Etienne-Emil Baulieu and colleagues recognized a role for progesterone (and other hormones) in central nervous system myelination. Over the next two decades, researchers uncovered the possible mechanisms and delineated more clearly for whom and in what types of injury progesterone seems most helpful. From studies of neurons (CNS) nerve cells (PNS), we now know that progesterone is key for myelination and neuron/nerve regrowth, at least in the acute stages. Progesterone stimulates myelination both directly by acting on oligodendrocytes and indirectly via actions on the neurons and the astrocytes that then message the oligodendrocytes to produce more myelin. Similarly in the PNS, progesterone aids in the remyelination and re-growth of nerve fibers, via the Schwann cells and via progesterone receptors located in what are called the dorsal root ganglia (DRG), the sensory neurons that carry information from the periphery to the brain. Whether in the CNS or the PNS, timing and length of progesterone administration are critical.
Animal Research – Progesterone, Nerve Injury and Neuropathy
The animal research has been mixed, but taken together, the results seem dependent upon the type of injury, the timing of the treatment and the methods of assessment. When treatment is begun early enough and extended long enough (this varies) and when the measure is neuropathic pain versus other potential outcomes (such as morphological changes to the nerve), there seems to be a favorable response. In rodents, single dose treatment does not seem to work, neither does treatment that is initiated too late after the injury or ended prematurely, though these criteria vary from study to study.
For example, using an induced model of diabetic neuropathy, researchers from Italy found that diabetes markedly reduced progesterone concentrations in male rodents within three months (females were not tested). This was the only study I could find that measured progesterone concentrations relative to treatment and outcomes. Chronic treatment (one month) with progesterone or one of its derivatives restored nerve function, increased key components of myelin production and reduced pain. Similarly, an induced model of trigeminal pain in male rodents found when progesterone was initiated early and at a high enough dosage, it tempered the experience of pain while increasing myelin producing proteins. Lower dosages did not work.
From Animals to Humans: Traumatic Brain Injury and Neuropathy
The research with animals, male rodents specifically, shows that progesterone treatment works best if given early enough, for long enough, and at high enough dosages. With acute or induced injuries under experimental conditions, early treatment is much easier than in real life where neuropathic pain develops much more gradually and often goes undiagnosed and untreated for some time. Would progesterone work in humans and would it work for chronic, well established neuropathy? The answers to those questions are not clear because the human research on progesterone and myelin focuses on acute injury, like the traumatic brain injuries. The human research also suffers from short duration dosing, includes mostly males, and without exception fails to address endogenous progesterone concentrations either pre or post treatment. Nevertheless, there are some indications that progesterone therapy may work.
Progesterone and TBI – Human Studies
In a smaller, single center open trial and two larger, double-blind, placebo-controlled, human trials, progesterone therapy was administered to individuals with severe traumatic brain injuries (Glasgow coma scale <8). In each case, the progesterone group did better, showed reduced morbidity rates than the placebo groups.
In the first study, 26 cases were treated with progesterone and 20 controls with placebo. At both 10 days and three months post injury and treatment, the progesterone treated group improved significantly more than the control group (abstract only).
In a second study, 159 patients, arriving to the treatment facility just eight hours post traumatic brain injury were randomized to receive either intramuscular injections of progesterone (82) or placebo at 1.0 mg/kg via intramuscular injection and then once per 12 hours for 5 consecutive days. Both intake neurological functioning and post treatment functioning were assessed and compared using a number of measures. Follow up assessment was conducted at 3 and 6 months post injury/treatment. The results were positive, albeit small. The progesterone treated group improved significantly across all measures showing consistently larger improvements compared to the placebo group. It should be noted that only 44 of the total subject population was female, 24 in the placebo group and 20 in the progesterone group. No analysis by sex was conducted and so it is not clear whether progesterone therapy works equally well in males and females.
In the third study, called ProTECT, a similar double-blind, placebo controlled, randomized methodology was used. Here, however, the randomization was 4:1 and favored progesterone treatment, whereas in the study cited above, the progesterone and placebo randomization was 1:1. Progesterone was given via IV for three days. The ProTECT study researchers found that patients in the progesterone had a lower 30-day mortality rate than controls (rate ratio 0.43; 95% confidence interval 0.18 to 0.99). While those who suffered more severe injuries had relatively poor outcomes at the follow up tests 30 days post injury, despite the treatment, and those who suffered only moderate traumatic brain injury and received progesterone were more likely to have a moderate to good outcome than those randomized to placebo (abstract only).
Two additional trials are on-going, hoping to test progesterone on thousands of patients: the ProTECT-III and SynAPSe studies.
Translating the TBI Research for Use with Neuropathy
What does improvement post TBI tell us about treating neuropathic pain from demyelination disorders? It is not clear, because even though researchers know that progesterone promotes myelination, the human research has focused narrowly on injuries where demyelination occurs but also where other factors are also involved in the outcome. We know from animal and cell culture research that progesterone attenuates the cascade of events that occur post TBI or post nerve injury via multiple mechanisms, inducing myelin regrowth is only one of those mechanisms. Progesterone reduces swelling of both vasogenic and cytotoxic sorts. It has anti-oxidant properties, upregulating enzymes that increase free radical elimination. Progesterone inhibits inflammation, stabilizes mitochondria, reduces neural excitoxicity and can limit apoptosis. Finally, progesterone promotes myelination. All factors that should point to consistent improvement in TBI and neuropathic pain syndromes, but the research is limited and mixed. Why?
The primary reason for mixed results is study design, almost all are short duration. Hormones are long acting molecules and the shorter duration may not be sufficient to generate the response, particularly when the injuries are severe or longstanding. Longer treatment regimes are likely in order.
Another reason for mixed results is the one-size-fits-all approach. None of the human studies and few of the animal studies, investigates why progesterone works in some subjects and not others. Almost all of the studies are predominantly male, rodent and human alike. None have investigated whether being female has anything to do with efficacy. None of the human studies measured circulating concentrations of progesterone, either pre-, during, or post-treatment and so there is no way to tell if those who responded had higher circulating concentrations or if improvement was contingent upon reaching a certain concentration.
Perhaps even more importantly, is the fact that progesterone, like any hormone, works within a vast and compensatory network of other hormones. The reductionist approach that utilizes a single hormone treatment protocol, while ignoring the potential cross-talk with other hormones and other variables is a consistent flaw these and other research protocols. Again, hormone measurement, progesterone and its metabolites, in addition to other key hormones, is imperative if one is to determine therapeutic efficacy.
I Have Peripheral Neuropathy, Should I Try Progesterone?
Progesterone therapy is generally safe, but as with everything there are risks. Women have been using it for generations in its bio-identical form to mitigate menstrual and menopausal symptoms. Since it is fat soluble, transdermal (skin) absorption is possible and progesterone creams have become popular. Some physicians prefer micronized progesterone, a pill form that reduces the molecule so it more easily passes through the liver without degradation. The pill form, and to a much lesser degree, transdermal progesterone, cause sedation and should be taken at night. Micronized progesterone has been shown to increase free thyroxine (T4) as well. For some women, and presumably men too, a gain of function mutation on the mineralocorticoid receptor can evoke very high blood pressure with any increase in progesterone concentrations (luteal phase of the menstrual cycle and during pregnancy especially). Although there are dosing references for progesterone relative to menstrual or menopausal therapy, the dosing is individualized and often includes the replacement of other hormones along with progesterone. Salivary hormone testing is used to monitor and hormone doses are adjusted regularly. Progesterone is also used predominantly for women. No such dosing considerations exist for men that I am aware of. Likewise, for peripheral neuropathy there are no references from which to design a treatment protocol and so it would be prudent to work with a functional medicine specialist, familiar with hormone management, to develop and monitor the course of treatment.
My Two Cents
I suspect, if progesterone therapy works for peripheral neuropathy, it will require a much longer term treatment period than is currently tested in the human trials. I suspect also, it will be difficult to ascertain whether it is the sole contributor to improvements in neuropathy symptoms, as neuropathy is a multi-factorial process that ought to be treated as such. Nevertheless, if you suffer from neuropathy and can find a physician to work with that is familiar with hormones and the research, progesterone therapy might provide a viable option, among other options like stabilizing thyroid hormones and supporting mitochondrial function.
Postscript
This article was first published February 19, 2014. Since then, a few more studies and review articles have been published and continue to support the role of progesterone in myelin regeneration, although the data are mixed. From a 2020 article.
Indeed, PROG and its metabolites modulate the expression of myelin proteins of the PNS, such as myelin basic protein (MBP), myelin proteolipid protein, glycoprotein zero (P0) and peripheral myelin protein (PMP22) as well as myelin formation [6,8,9,20,63,64]. In particular, the expression of P0 in the sciatic nerve of adult male rats, as well as that in Schwann cell culture, is increased by treatment with PROG, DHP or THP.
…Data here reported support the concept that neuroactive steroids, synthetic ligands acting on their receptors or inducing their synthesis, may improve PN symptoms, including neuropathic pain and consequently may represent an interesting possible therapeutic strategy. In addition, based on the sexual dimorphism of neuroactive steroids as well as of PN here discussed, a gender specific treatment based on these compounds may be also proposed.
We Need Your Help
More people than ever are reading Hormones Matter, a testament to the need for independent voices in health and medicine. We are not funded and accept limited advertising. Unlike many health sites, we don’t force you to purchase a subscription. We believe health information should be open to all. If you read Hormones Matter, like it, please help support it. Contribute now.
Yes, I would like to support Hormones Matter.
This article was published originally on February 19, 2014.











Hi there, My Mom is 76 and had a terrible accident more than a decade ago. Since then seh’s been sufferung from terrible neuropathy. Do you think that progesterone could help here – at least a bit?
At this point, maybe not. I believe it is used fairly soon after the damage. What you might consider, however, is thiamine. Most older adults are deficient and require more thiamine than is required by younger adults because of problems with absorption. Take a look at the articles on the site regarding thiamine, you might it quite useful. Note, thiamine requires magnesium to activate, and again, most folks are deficient. Good luck.
Estrogen regulates tryptophan metabolism by acting on an enzyme that shifts away from the formation of niacin, towards excitotoxin quinolinic acid. Progesterone inhibits the activity of this enzyme.
y en hombres como sería?
I’m glad to have found this article, which has confirmed my suspicions about the effects of low progesterone on my current condition.
I have PCOS, which requires hormone therapy. Two years ago I suffered some pelvic trauma, which left me with minor irritating nerve pain. I have also been taking bio-identical progesterone for a year (200mg two days each month). I went off of it three times as my doctor suggested when it wasn’t stopping my menstrual bleeding like it was meant to (I have menorrhagia).
I began having Vulvodynia last year. Three flare-ups all together so far. Things down there would swell and turn red. Then go away after two months. I was able to tie the flare-ups to the times I went off the progesterone.
Sadly, I am having trouble finding a doctor that will proscribe these to me, as there is a fear of them causing cancer. European women have been taking these things for decades without problems in the majority of the women and I am not at risk for cancer based on my family history. It helps with my mental health too, so it’s very infuriating. My very life is in the hands of those that lack information or the desire to learn.
Really interesting article, it never fails to amaze me what progesterone can do for us. Synthetic progestins, that is in HRT and birth control is completely different from natural progesterone and most of it gets lost in the liver and gut if taking it orally. I love what natural progesterone has done for me, especially elimination of menopause symptoms. I find it easiest to buy online. For the last 3 years I have been using natural progesterone cream, with paraben free, organic ingredients. Website I use if of help to anyone –
progesteroneorganic.com
I am 40 years old female, married and have one daughter of 10 years old. I am suffering form chronic bacterial vaginosis and peripheral neuropathy for last 2 years (all these symptoms developed after a failed IVF). I went to many gynaecologists and tried many different types of Antibiotics (both oral and suppository), I tried both provacare and probalac probiotics but nothing worked for bacterial vaginosis. I also went to many neurologists for my feet and hand burning but they just suggested some B vitamins. I am taking Neurobion and Pregabalin regularly but it is not helping that much for my feet burning pain.
Can you please help me? The burning pain in my feet and hand is seriously affecting my life.
That is more likely related to the mitochondrial damage done by the IVF drugs and the subsequent antibiotics. Take a look at our posts on mitochondria and on thiamine. I suspect you’ll find your answers there.
I stumbled across your article after a recent pregnancy when I was looking for links between sex hormones/pregnancy hormones and decreased neuropathies. I am 26 and was born with fully closed meningocele Spina bifida (L3,L4 mostly). I have herniated disks and recently went to my neurosurgeon for an MRI read. He said all things considered it looked good and that it’s likely that the pain I experience is just something I’ll have to deal with. I’m not buying that there’s not something out there that can at least minimize the pain other than narcotics. I’m fortunate to be able to walk but nerve spasms in my legs have become debilitating throughout my life. They are much more frequent and intense off of my birth control and around the time of my period and ovulation. Unfortunately this pregnancy ended is miscarriage but I couldn’t help but notice for the time that I was actively pregnant, my nerve pain (sharp electric spasms) went from daily/hourly to having about 2 pains in a 4 week span. I made a comment to my husband about how wonderful I felt. Since miscarrying my pains returned and are the exact same. I can’t help but wonder if the increase in progesterone production and LH hormone decreased my pain and if something in the future could be taken to act the same way. Do you have any input on this?
I am experience similar issues. I was feeling fabulous until I woke up one night 6 months ago feeling pressure on my shoulders, numbness of my left jaw, left arm and leg. Went to ER and they found I had cervical radiculopathy. Followed up with primary who recommended seeing a neurologist. I had been going for physical therapy as well. The symptoms were strange and moved all over my body not just one side and included numbness slight weakness/rubbery feeling in my lower arms, tingling, electric shock feelings and twitching all over my body. Had MRI and MRI with contrast with showed an arachnoid (sp?) cyst and slightly enlarged pituitary. Saw primary, endocrinologist, ortho and gyno; had all bloodwork come back normal and they were not concerned with cyst or pituitary. I also realized that I noticed symptoms of peripheral neuropathy including burning tongue, along with above symptoms that come and go. I’m taking gabapentin which has helped somewhat but I still feel a sensation on my feet and lower legs and also have lower leg pain. The Drs all say that all these symptoms are not caused by radiculopathy. I’ve been going in circles from one dr to the next who just keep sending me to others. After reading this I’m thinking hormones could quite certainly be the culprit. I’m 53 and not officially in menopause (haven’t gone full year without period). I will try the progesterone cream and ask my dr about other therapies. Any comments/suggestions welcome – this has been a frustrating and frightening experience. I feel for you all who suffer 🙁
Hi, I’m from Australia… born in the US…lived in Australia over 50 years…I’m 54 have had 5 children, married to an Aussie. We live on a farm and I love life.
About 8 years ago I started to have symptoms of PN…I have Cervical Spine degeneration(OA) and L5 disc bulge(done over 20 years ago). I went to a neurologist as I was experiencing migraines and a CAT scan revealed possible demylination in the brain…I have a cavernous hemangeoma in the cerebellum but it has not changed at all and is of no worry. I was also given a spinal tap which came back with positive changes to the ongogloncal bands and was told I had MS. It was suggested I’d be in a wheelchair if I didnt slow the MS process and take Galenya, but I thought differently so every year I have a follow up MRI with no brain changes and still have the PN…it is increasing. I had a hysterectomy in January this year due to very heavy periods…so far it has been a success, but I am in menopause.
Your article intrigued me about Hormones and I’d love to know more about your work and if you know of any doctors who follow your thoughts here in Australia.
My other reason for wanting to know more is because of my husbands recent farm accident…On March 13th he went out to check on something just on dark and tripped backwards hitting the back of his head on the excavator chassis…he has sustained traumatic brain injury and was in icu for 2 weeks…we didn’t think he would survive because the left frontal lobe was damaged too…cut a very long story short he is walking talking and has recovered so well…will not be 100% but it is clearly amazing. I was wondering if hormone treatment may help in his recovery? He is 61 y.o.
Looking forward to hearing from you…
Thanks
Ruth
I have a condition called Charcot Marie Tooth & have severe nerve damage and pain. I noticed while on HRT in a Combi-patch the pain in my feet calmed somewhat.
My GP is not writing my prescription anymore and being off the patch for over 2 months the pain is becoming unbearable! This led me to research this subject.
I’m a 41 year old woman that has been suffering from an L5S1 disc herniation for the last 10 months. I have had 3 surgeries to try and “fix” my nerve pain. My first surgery removed my left ovary and Fallopian tube as it was covered in an significantly large abnormal cyst and they thought that could have been pressing on my S1 nerve root causing he pain. It did nothing to address my radiculopathy and after several Conservative efforts, I ended up having 2 separate micot discectomies on the same L5S1 disc herniation. The doctors have thrown all Kinds of narcotics at me and 8 rounds of prednisone in just 6 months, nothing seems to help. My nerves continue to react in my one injured leg with burning, tingling, ripping, crushing, searing electric pain and twitching (this list goes on but I will leave it at that), but now 3 weeks post 3 rd surgery and muscles all over my body now with twitching too. I know my pain is always more intense during my monthly cycle and post operatively I’ve had gobs and gobs of hair falling out, just like after delivering my babies. I have massive amounts of hair, so this doesn’t seem to be a negative thing at his point, I just feel like my pain is so closely associated with my bodies hormonal cycle and I want some answers, not just to hear “it’s normal try this oxy and prednisone and see us back in a month”
In addition to the structural issues you may have, you may also have significant mitochondrial damage because of all the medications. Hair loss is an early sign of mitochondrial damage, usually corresponding thyroid damage as well. So too is is the electrical sensations (of course, barring obstructive damage to the nerve from the surgery). And in fact, since steroid hormones require healthy mitochondria, so too are the hormone issues. Read up on mitochondrial health on our site. If you can heal your mitochondria, mainly through nutrients and diet, that’s all they need, the mitochondria will kick back in and begin to heal the nerves and some of the other issues you are having.
I am 47 and in menopause. I was put on the combipatch. I was only on it for 8 weeks when I started getting all over numnbness and burning in my hands and feet. Went to neurologist and she told me to remove patch that it was coming from it. I also suffer from complicated migraines and low thyroid. I want some relief. Any suggestions would be most appreciative.
I’m in the UK and have had a full hysterectomy, with bilateral oopherectomy. I’ve been having problems with nerve pain down the whole of my left side, which is made worse on the higher estrogen dose (which I need, because I’m in surgical menopause at a young age).
I’ve begged with doctors and consultants to try progesterone, but every time they say no, as I have no uterus! Is there anywhere in the world that would consider prescribing progesterone for me to try?
Thank you.
In the US, you can get progesterone creams over the counter but more potent ones at compounding pharmacies. I think also that you can order these online without a prescription, but I am not sure.
Even if you were to get “progesterone” from a doctor in the UK, there’s a big risk that it wouldn’t be progesterone at all, but rather any one of several members of the progestin class of hormones (progestins are manmade substances that are designed to mimic the action of progesterone). Despite what doctors might try to tell you, progestins are not progesterone. In particular, progesterone increases neurosteroid synthesis whereas progestins have the opposite effect of depleting neurosteroids, and are likely to make things like peripheral neuropathy worse rather than better!
Fortunately you can buy progesterone cream online in the UK without a prescription. One way is by ordering from the US amazon.com website, or direct from online vendors in the US (most of whom will ship to the UK). The drawback of that is long delivery times, and also there’s a big risk of being hit with VAT, and post office VAT collection charges, which end up more or less doubling the cost of the order! There are also two UK-based online vendors I know of that sell progesterone cream, biovea.co.uk and onasnatural.uk.
In the US too if you go to a conventional doctor most will suggest a progestin. Few seem to know the difference. We have compounding pharmacies that specialize in bioidentical hormone therapies, so we can get progesterone and other hormones fairly easily in personalized doses and I think the progesterone cream is over the counter.
I went off the birth control pill when I was 42. I went straight into menopause, hot flashes-the whole thing was awful! I went on HPT and within 6 months I began to feel tingly in my feet and low legs, the neuropathy got worse and over the past 8 years it has become unbearable. I am on my 3rd neurologist, and have no sensory in my feet or low legs, you can poke me with pins and I can’t even feel a thing. I have done many treatments to help, the Calmare treatment, a year of IVIG infusions that didn’t really help, and am on an anti-seizure medication. I even had a spinal tap. I now believe that this is all due to a dump in my estrogen/progesterone levels 8 years ago when I went off my birth control pills. The HPT levels are not near what they were when I was on the pill and for whatever reason I think maybe I just need a higher level to produce more serum in the dorsal root ganglia area to protect those sensory nerves. So at 51 years old I am contemplating going back on the Birth control pill, I know there are higher risks of breast cancer and stroke, but I just want to be able to walk without pain again. Do you have any thoughts for me?
Have you tried progesterone creams?
Given your age, I would highly recommend against using birth control again. The stroke risk would be too high.
I would also consider looking into diet and nutrient deficiencies. Thiamine deficiencies, in particular, can cause neuropathies. Declining thyroid hormones too. Finally, have you ruled out disk herniation and/or pinched nerves.
This discussion is a beacon of light for me. Thank You Dr. Marrs! I’m a 40 year old RN and just discovered that I have Estrogen Dominance. For the last 2 years I’ve endured myriad of symptoms that can only point to low progesterone levels. I’ve felt quite awful, had a miscarriage, had terrible PMS, painful breasts, spotting in between periods, depression, anxiety, numbness and tingling and in my arms and legs, and neuropathic pain in those areas as well. I just started progesterone cream and I’m looking forward to the possibility that my peripheral neuropathy will finally be cured. After a full neurological work up there is no other explanation and all signs point to my lack of progesterone. Fingers crossed. Thank you for a wonderful and informative discussion!
You’re welcome. Let us know if it works.
Hi. Did your neuropathy improve with Progesterone cream.
what about men testosterone? I know that progesterone is important for men too, but can a man lack it?
Just wondering if you were ever helped by the progesterone cream? I have been experiencing the same symptoms as you, minus the depression and miscarriage. I have been through literally every test there is trying to determine what is causing my anxiety, and numbness and tingling in my left cheek, left hand, and left calf, which started about 10 months ago. Everything comes back negative, thankfully! The other PMS symptoms you mentioned I have also lived with since puberty. I am now in perimenopausal stage, so my current gyno feels it might be worth trying the cream. Any luck with it? Thanks!
Hi,
I had painful bladder syndrome (actually not the official diagnosis in the 80s, and was operated on for it. Ovary removal within the next year following. Painful bladder went away until I turned 49. I had a few intermittent episodes lasting a brief time on a few occasions during that year. I had a total hysterectomy the year I turned 50. I actually felt pain during surgery and have suffered from nerve pain ever since. I have been diagnosed with PN. I found it more than coincidental that the bladder pain was during periods where my hormones were changing. Also, I have often wondered if hormones were playing a part in this. Since there are very few doctors that address pelvic pain, it has been a nightmare dealing with this and figuring out what to do. I’ve considered going to a hormone center that tests for everything (I’m also losing my hair and yet, have the dreaded facial beard (exaggerating of course). I was diagnosed with hashimotos around 47 years of age, but my blood work comes back normal so I am not on any thyroid medicine. I’ve been suffering from PN since my surgery nearly 4 years ago. Is it too late for me to benefit from progesterone. Please advise. Thank you
My husband has diabetes and neuropathy. I was looking into progesterone for a friend with M.S. and thought why not try it for my husband’s neuropathy? I ordered the Emerita cream and he uses 1/8 teas. at night. After only a few days, he said the burning and tingling have almost disappeared.
To avoid buildup in fat cells, the cream should not be applied to stomach or buttocks. Alternate areas each night between inner wrists, neck, face, upper chest, back of knees (places with lots of blood vessels and little fat).
I am hoping my friend will try it for her M.S. and see some results. She noticed that the M.S. symptoms came on around menopause when progesterone is very low in women. Who knew it could work for men too??
It’s time to do our own research and not wait for our doctors to wise up. Thank you for your web site and wisdom.
I stopped taking a lose dose of Premarin because my insurance no longer covers it. I am 71. I had tried stopping it several times but the hot flashes were so bad that I continued again. Now I have neuropathy in my feet–a constant burning. Can this be connected?
Hi,
My mother is 68,in the past two years she developed peripheral neurphaty. she had been using ACTIVELLE (NOVO NORDISK A/S, DENMARK) for 10 years because she felt better using it scince premenopause. Now a few doctors recoended to stop using it because it might be the cause for the developed neurophaty. My mother also had hypothyroidism but she is using Levothyroxine Sodium. since she stoped the hormones the neurophaty and general feeling is worser…do you think that Activelle might be the cause for the neuropathy? do you think that it is safe for here to go back using Activelle? or would you recomand other things?
I developed multiple symptoms 1 1/2 years ago after having influenza A and being on prednisone (on and off for a couple months). My hormones are a mess and I have had neuropathy believed to be PN. It is painful, ugly and seems to be exacerbated by hormonal fluctuations – I am 52. No one has been able to help and I am living in pain on a regular basis. The pain seems to be worse directly after my period. I really need help. I have all the symptoms, burning, tingling, numbness, twitching muscles, stabbing pain.
I would consider the possibility of progesterone but also look into thiamine deficiency, hypothyroid and vitamin B12 deficiency, they seem to all go hand in hand. We have a lot of articles on these topics in the archives, please dig around and see if anything fits.
My mother suffers from TN and my husband has been suffering with neuropathy – could life be as simple as trying them both on progesterone? What a wonderful thing that would be. Please share any and all tidbits, you never know what little fact could possible help another person, in this case two!
Hi…I’m 45 and My “medical mystery journey” started 6 months ago with tingling/cold lower extremities. I have seen 2 neurologists, had 2 MRI’s, have seen a spine surgeon, my endocrinologist (as I had my thyroid removed 16 years ago and am on Levoxyl; a naturopath (who is currently treating me for an inconclusive Lyme test.Lupus, MS and Diabetes have been ruled out. Nobody knows what to do with me 🙁 Legs have been really tingly and cold the past few days. Really want my life back 🙁
AND my hairdresser discovered Alopecia (bald) spots recently. I now have 3. Two on the top of my head and one on the back of my head. I’m a mess 🙁
Consider looking at your thyroid. Have them test for anti-thyroid antibodies as well as T4, T3. We have articles on hair loss as well neuropathy on the blog. The thyroid is generally involved as are nutrient deficiency – b12, B1 – thiamine, being the most prominently linked to demyelination – the underlying cause of neuropathy in many cases.
Hi Marianne, My symptoms are very similar to yours……
I’m curios to learn how you are getting along ? I too have got freezing cold lower legs….but not my feet. I have some tingling in my finger tips, but its random. My upper thighs on the back side freeze and so to my buttocks. I am 50yrs post menopause, no VitB or VitD deficiency. My GP is at a loss and I’m waiting to see a Neurologist next week. I have just started HRT.
Hi Sharon…I’m still miserable 🙁 Legs still frozen/burning from the knees down (including feet). Am waiting to see a neurologist in Boston. How are you?
I am having the same thing now.. i have been tested for everything you have and still no diagnosis – did you ever find out what it was.. I am seeing this 2 years after your post
At age 35, I developed epilepsy which appeared to be catamenial. At age 41, I became pregnant and the elevated progesterone to estradiol ratio seemed to stop the seizures which had been previously refractory to a number of anticonvulsants. Following delivery, my seizures sky-rocketed and I was put on progesterone cream. I was diagnosed at age 42 with “mild Addison’s Disease,” due to the fact that one adrenal and kidney were removed at age 9 due to a pheochromocytoma. The remaining adrenal is intact, but DHEA and testosterone were found to be deficient. My endocrinologist prescribed replacement adrenal hormones: progesterone, DHEA, hydrocortisone and Florinef. During peri menopause, seizures got worse despite treatment with 100 mg/d progesterone and multiple anti convulsants. Post menopausal, seizures continued even though adrenal hormone, and anticonvulsant treatment continued. At age 52, I developed peripheral neuropathy in feet. Mayo clinic says it is idiopathic, axonal sensory neuropathy. At age 57, I started neurofeedback for seizures. At age 60, I have been seizure free for 18 months thanks to the neurofeedback. Neurofeedback has not helped with neuropathy. Any ideas about how my hormone regimen (progesterone, DHEA, hydrocortisone and Florinef) might affect the neuropathy? Thank you.
Low T3 (common in hypothryoidism/hashimotos), will induce neuropathy as will low vitamin B1 (thiamine) and low vitamin b12. I would check all three. We have articles on these as well, some of them are relative to the individual deficiencies, some relative to different medication/vaccine adverse reactions (many meds/vaxes attack the thyroid and deplete a multitude of nutrients that are responsible for mitochondrial function and myelination); begin there for more information.
Hi, I have deep bone pain which started after my son was born. Doctors are not able to find what is wrong but i get the pain during ovulation and every time i fall pregnant. I’ve had three miscarriages in the first 3-5 weeks and get the same pain but it’s amplified. What could this be? Thanks.
That’s very interesting. I don’t know what it could be, but if it is consistent with ovulation and pregnancy, it is likely hormonal; which hormone and by what mechanism, I am not sure. Do some digging online, you may find some research. If you do, please share and I will dig further.
Hello, I was taken off estrodial about 18 months ago. I have had fibromyalgia for many years and take Tramadol daily. In sept I found out I now have Hashimoto’s disease. My thyroid was fine a year ago. Having horrific pain in hand arms feet calves and it spreading. Also unbelievable hot flashes. I’m not over weight and hv always been active. Right now walking is unbearable I am going to ask to be put back on progesterone and estrogen tomorrow. Dr stopped me from taking it when I turned 63. Anyone else have this complaint. I’ve found it very frustrating trying to get relief since August.
For the last couple of months I have been waking in the middle of the night/early morning with a numb hand on one side or the other. I suspect Hashimoto’s Thyroid but haven’t made it to the Dr yet. I did notice that when I got my period last month the numbness went away for 5 nights in a row. Last night was the 1st night since then that I haven’t been awaken by numbness. Low and behold I got my period this morning. Does your progesterone surge while you menstruate? I am 44 so I am approaching menopause so there are all kinds of hormonal things going on! Great article, something to think about & look into!
I took myself off progesterone in June 2015 (using it for hot flashes). I am just now putting 2+2 together, but a neuropathy that had been stable and very minor for a decade has gone SEVERELY downhill to the point where it’s my hands ANd legs AND feet, like I’m wearing knee socks full of hot lava. Frightening. I just begged my doctor to let me go back on it if it will at all help this horrifying, frightening problem.
This is fascinating. Why won’t the doctor let you go back on the progesterone? Were you having other side effects?
Re:progesterone and peripheral neuropathy
I am being treated with 100mg progesterone as indicated by blood test results. The happy spin-off is that my trigeminal neuralgia has cleared.
I been suffering with Trigeminal Neuralgia since 9yrs. My episodic attacks started during peri-menopause yrs. After I started on BHRT they become less severe and finally I’m on remission. Each time I stop BHRT my pain comes back. I’m on 100mg Progesterone and 0.0375mg Estradiol Patch.
Hi Susan, are you also still free from the TN pain?
Hello Susan,
I have TN as well, also started at the onset of peri-menopause, about 5 years ago. I am so glad for you the progestorone and estradiol helped you so much. I think I have read you also on ALA and Myelin sheath repaer? I want to ask you what helps you the most? Thank you so much for sharing, you are a big help to me just by sharing your story that is simular to mine.
Greets and thanks again,
Mary.
Very interesting. I have had TN now for over 5 years. It had began about 8 months after I had an IUD inserted and was very intense for about 6 months. Again I am in intense pain 8 months after having IUD replaced. Too coincidental! At the time I had the IUD replaced I asked specialist if my TN could have been caused by hormones and was told no way. Going to get it out as soon as I can and have started the process of getting hormone levels checked.
Hi Ellena, Are you still free from the trigeminal neuralgia pain?
Do you think it is because of the progesterone?
Hi Ellena, I saw what you wrote and wantes to ask you What kind and What Form of
Progesterone has Helped your TN
I have been Suffering so Much, I was
just about to try some Progesterone
lately. My name is Diane and it wou
be Great to talk with you, I hope we csn.
This is my E-mail – logcreek18@yahoo.
and my Number, 860-995-4825.
I really hope we can Talk.