Introduction
Magnesium is the most common divalent metal cation present in mammalian cells. It is estimated that the average 150 pound adult stores approximately 24 grams of magnesium in body tissues. Magnesium is an intracellular ion with relatively high cytosolic concentration in the millimolar range. Up to 99% of total body stores of this mineral reside either in bone (54%) or intracellularly (inside of the cell – 45%) – wherein skeletal muscles and soft tissue harbor most of the intracellular component (2). The magnesium inside the cells is mostly bound to cellular molecules and machinery that include proteins, enzymes, phospholipids, and deoxyribonucleic acid (DNA), as well as energy molecules including adenosine triphosphate (ATP). In this way magnesium is set to activate the metabolic enzymes and cellular machinery in the different cell compartments, as well as promote ATP production and activation – ATP can only be optimally functional when bound to magnesium.
Magnesium Deficiency and Testing
The National Health and Nutrition Examination Survey (NHANES) surveyed Americans’ intake of magnesium between 2005-2006. The conclusion was that almost 50% of Americans have insufficient intakes of magnesium from food and water. This means that millions of Americans do not consume the Recommended Daily Allowance (RDA) of magnesium (310-420 mg – depending on age and gender). It is important to note that these minimal numbers are required in order to avoid overt signs of disease and they do not imply optimal levels needed.
It is difficult to accurately measure magnesium levels as currently there is no one cost-effective and easily-available test that can reliably predict magnesium stores. Serum or plasma magnesium levels do not accurately predict total magnesium tissue status. This is because plasma levels of magnesium account to about 1% of total magnesium. Plasma levels are balanced with magnesium from bone and tissues. What this means is that the body stores can become substantially depleted, yet the plasma levels being tightly regulated may not move in terms of magnesium concentration giving the wrong idea that the person is not deficient. Overall, plasma should be thought of as the body’s “highway” transportation system and in some cases, will not reveal deficiencies hidden within tissues.
At the moment, the best test for predicting magnesium deficiency, in terms of cost effectiveness and accuracy, would be the red blood cell (RBC) magnesium. This test can be ordered through your physician or via online services which will provide you a diagnostic laboratory requisition so that you can get drawn and tested. The RBC magnesium test is much more accurate than serum/plasma magnesium levels in predicting magnesium stores. This is because it has a larger test dynamic range and the numbers given by this test tend to move up or down based on how much magnesium a person may be ingesting or if the body stores are low. Many providers use this test to monitor therapeutic dosing of patients.
Magnesium and Immunity
Magnesium is not typically viewed in context of immunity. As important as it is in ameliorating symptoms of chronic syndromes that include insomnia, anxiety or fibromyalgia – it is actually an important immunomodulatory factor. The following discussion herein will focus on magnesium in context of immunity. The role of magnesium in controlling the arms of the immune system has been known since at least the 1990’s. These include cells of the innate immune system, such as macrophages, as well as cells of the adaptive immune response including T lymphocytes.
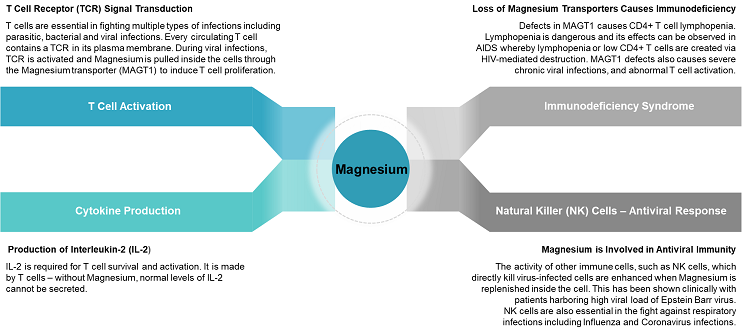
The two arms of immunity work together in a complex and coordinated fashion in order to monitor, detect, and initiate responses against foreign invaders. The innate response is the first line of defense and is genetically pre-programmed to detect variety of common patterns or chemicals present in viruses, bacteria, and parasites. The adaptive immune response, in turn, is “specialized” and is able to modify its genes in innumerable combinations in order to create extremely specific agents, such as antibodies, that will find and lock on invading pathogens. The adaptive immune response is dependent on the innate system for reporting invading pathogens. In contrast, the innate immune response will typically undergo the first battle but then will depend on adaptive immunity to secure long term protection from that same invader.
X-linked Immunodeficiency with Magnesium Defect, EBV infection and Neoplasia (XMEN)
One of the most prominent demonstrations regarding the role of magnesium in immunity occurred with the discovery of the XMEN syndrome in 2011. The group, led by Dr. Michael Lenardo from National Institute of Health (NIH), demonstrated detection of a magnesium transporter defect that results in CD4+ T cell lymphopenia (i.e. low number of specific T cells). The defective protein, called MAGT1, is present on the X chromosomes (hence XMEN). This makes it more likely that males will present with symptoms as females have two X chromosomes and can compensate with the second normal copy of the MAGT1 gene.
MAGT1 is a magnesium selective transporter which is present in immune cells – in this case it would be T cells, which are part of adaptive immunity. As discussed briefly above, adaptive immune cells have the ability to re-arrange a portion of their DNA in order to create cell receptors that can identify numerous pathogens. Once T cells encounter their cognate pathogen they will be activated.
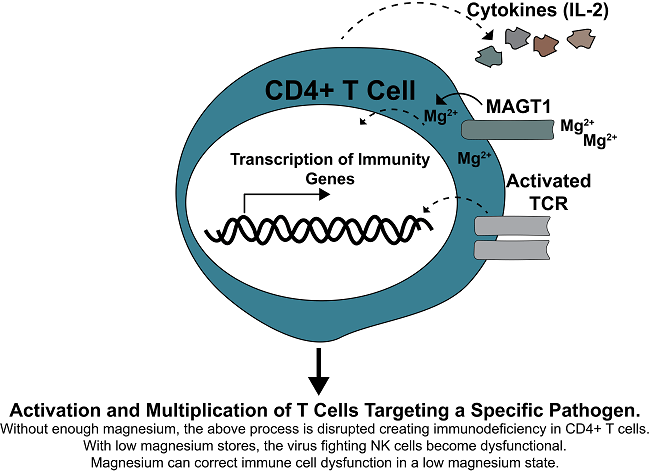
Activation of T cells occurs when the T Cell Receptor (TCR) is activated during infections. Optimal TCR activation requires highly coordinated interactions of numerous and diverse array of enzymes that are activated in a web of signaling interactions. Some of those proteins that are activated allow release of magnesium inside of the cells. What Dr. Lenardo and his group found is that without functional magnesium transport, enzymes that subsequently allow calcium in the cell are not properly activated. This in turn causes major defect in T cell activation and interlukin-2 (IL-2) production (3, 4). IL-2 is a critical cytokine that T cells produce in order to increase survival and multiplication of pathogen-specific T cells. The importance of IL-2 can be observed in the pharmaceutical world where IL-2 mechanisms are directly targeted to promote immunosuppression in individuals undergoing organ transplantation.
MAGT1 deficiency here clearly demonstrates the effects of not enough magnesium inside of immune cells. Magnesium is so important that its deficiency (i.e. MAGT1 dysfunction), in some ways, will clinically resemble AIDS patients that have very low CD4+ T cells due to HIV-mediated destruction – that is, susceptibility to variety of infections.
Magnesium Corrects Defects in Interlukin-2-inducible T Cell Kinase Deficiency (ITK)
A recent study demonstrated another fascinating display of correcting magnesium-related immunodeficiency. In this case, a patient presented with another genetic defect (ITK deficiency) that has similar clinical symptoms to MAGT1 deficiency discussed above. Here, the authors administered magnesium to the patient’s cells in-vitro (i.e. in cell culture) and found that magnesium restored the function of cytotoxic CD8+ T cells. This observation, combined with MAGT1 correction in-vivo (patients supplemented orally) demonstrated the importance of this mineral to the function of immune cells.
Epstein Barr Virus and Innate Immunity
Epstein Barr Virus (EBV) is member of the human Herpes virus family that includes Cytomegalovirus (CMV) and Human Herpes-virus 6 (HHV6). These viruses are associated with immune dysregulation, and Chronic Fatigue Syndrome (CFS) – also termed Myalgic Encephalomyelitis (ME). Loss of a magnesium transporter MAGT1 in immune cells, not surprisingly, coincides with increased levels of EBV in blood. High chronic levels of EBV predispose one to lymphoma. Part of the reason behind unchecked levels of EBV in MAGT1 deficient patients is the dysfunction of another arm of the immune system, Natural Killer (NK) innate immune cells. As the name implies, these “natural” killer cells are programmed to find infected cells (as well as cancer cells), lock on them, and then literally fire cell-killing material at them causing their obliteration. However, in the case of MAGT1 deficiency these NK cells become non-responsive and unable to mediate clearance of EBV. Importantly, magnesium supplementation in these patients corrects the impairment in NK cells and allows clearance of EBV-infected cells. From these studies we can speculate that chronically low magnesium levels are likely to promote a spectrum of immunodeficiency similar to the gene dysfunction seen with MAGT1 protein.
Respiratory Viruses
NK cells are involved in clearance of numerous viruses, including respiratory viruses. In fact, Sorrento Therapeutics is studying NK cell based therapy for treatment of Coronavirus 2019-nCoV (COVID-19). Dr. Robert Hariri, CEO of Cellularity (partner of Sorrento Therapeutics) in a recent interview states
“Not everybody exposed to COVID-19 gets sick. That suggests that there is something fundamental about them that makes those who get sick and those who don’t get sick different – and we know that’s their immune systems.”
This is paramount, as one of the known modulators of immunity, specifically with NK cells, is magnesium. In essence, to have optimally functioning NK cells for overcoming viruses a person must have sufficient amounts of this mineral.
Therapeutic Dosing of Magnesium
In order to replenish already low magnesium levels one must undergo dosing (under medical guidance) for at least 1 month and then re-evaluate levels. As mentioned, magnesium RBC is currently the most cost effective with acceptable accuracy. In a study performed by Jigsaw Health, magnesium RBC levels were significantly increased by 30% within 90 days of oral supplementation. This coincided with substantial improvement of magnesium-related symptomology – i.e. symptom decrease of 63%.
There are several options of magnesium compounds, some of quality and others more or less useless in context of mineral repletion. In short, some of the best forms of magnesium compounds are the chelated molecules. This includes magnesium glycinate and magnesium malate. Additionally, magnesium chloride (known as “oil”) is an effective remedy for local application but can also be used internally.
There are several resources on which the reader can further entertain this subject. The list includes, Dr. Carolyn Dean (author of The Magnesium Miracle), Morley Robbins (author of the Root Cause Protocol), Dr. Mark Sircus (author of Transdermal Magnesium Therapy), and Thomas DeLauer (Fitness Expert). These sources delve into other physiological effects of magnesium not mentioned on this discussion which is centralized on magnesium-related immune responses.
Summary
Magnesium is an essential mineral and is required for numerous enzymatic reactions. ATP-utilizing enzymes require magnesium as a cofactor. Without this, the cellular machinery and metabolic functions will be substantially reduced. Magnesium levels can be increased in the cells and tissues with chelated or liquid forms that are generally safe to administer.
Immunodeficiency stems from several factors, including environmental and genetic factors. We now know that deficiency in a magnesium transporter protein, MAGT1, found on the cells’ plasma membrane results in immunodeficiency termed XMEN. This deficiency results in AIDS-like syndrome with lymphopenia and CD4+ T cell deficiency. Additionally, this deficiency creates defects in NK cells, which predisposes the carrier to multiple viral infections as well as increased risk of cancer. The latter is due to reduced tumor-surveillance mediated in part by NK cells.
Repletion of magnesium in genetically compromised individuals allows for correction of some of the presented defects. We can deduct from these findings that persons with low magnesium stores, but not genetically compromised, will have a higher chance of being in a spectrum of immunodeficiency, with chronic depletion probably causing overt symptoms. However, more studies need to specifically define what level of magnesium deficiency will correlate with immunodeficiency observed in XMEN syndrome.
We Need Your Help
More people than ever are reading Hormones Matter, a testament to the need for independent voices in health and medicine. We are not funded and accept limited advertising. Unlike many health sites, we don’t force you to purchase a subscription. We believe health information should be open to all. If you read Hormones Matter, like it, please help support it. Contribute now.
Yes, I would like to support Hormones Matter.
This article was published originally on March 23, 2020.














Having a cardio-renal situation, I am wondering if the recommended magnesium taurate is safe at levels that are normally used in making up for Mg deficiencies. I’ve read where “excess Mg taurate levels can be nephrotoxic” at the levels considered “excessive”. Do Mg salts have problems with individuals with CKD? Articles that use terms like “excessive levels of Mg” mean little unless there are actual numbers being applied to these situations. Dr Lonsdale favored usage of Mg taurate and I ask how safe levels can be applied to deficiency situations. Otherwise, it sounds like a nice compound as both taurine and Mg are very valuable to humans.
Hello Michael,
Although hypermagnesemia (too much blood magnesium) is a frequent complication of renal failure, it doesn’t necessarily imply that individuals with CKD should not have magnesium. On the contrary, given appropriately, magnesium supplementation reduces complications that may come with CKD like vascular disease or abnormal bone-mineral metabolism. A strategy you can take to your doctor for consideration is dosing magnesium salts first, or something like magnesium citrate at low doses in appropriate intervals. Then ask your doctor to monitor your blood level magnesium on your checkups so that you know how the dosing is working. If all works alright then see if your doctor is willing to try out magnesium taurate with monitoring of blood.
Does “blood level magnesium” refer to Mg in the RBC’s or blood serum or both? Isn’t serum magnesium tightly maintained whereby about 1% of total Mg is in serum? Previously, Dr Lonsdale said he liked Mg taurate preferably and I am curious as to what concentration/total mass of Mg would be considered nephrotoxic? Taurine is split off from Mg taurate and becomes taurine. Have toxicity curves been generated previously plotting concentration versus some index of kidney injury? Would you expect Mg taurate to be more nephrotoxic than any other salt of Mg? Dr Lonsdale has said that Mg taurate is more effective than the combination of taurine and the most common salt of Mg. Taurine sounds like it is valuable in treatment of heart failure and has been used for 35 years in Japan for heart failure to good effect.
In this case it would be serum magnesium, but yes in the typical case you want Magnesium RBC for a better indication of total tissue levels. It would not be a bad idea to also add RBC mg just to monitor the efficacy of treatment. So use serum levels to make sure hypermagnesemia does not occur and RBC for efficacy of absorption into tissues.
I have not looked into studies demonstrating quantitative toxicity curves of taurine in patients with renal failure. Although studies do seem to mention benefits of taurine to the kidneys. This study for example shows a positive correlation between taurine excretions (i.e. intakes) and the survival of renal grafts (after transplantation). https://www.mdpi.com/2072-6643/11/9/2212
The reason I mentioned salts/citrates is because they are published for use in patients with CKD – though there are likely studies out there using taurine. The idea is to go with the citrate route first and then commence on taurine once you and your doctor see that the first variable is stable (magnesium).
What do you mean by the term “kidney failure”? Does it mean that one is either on dialysis or dying? If one has level 3a kidney disease, classed as “moderate”, is this the same as one in total failure or not? To me, failure means kaput. Kidney disease need not always signify the patient will soon die.
Some time back I read that there is a certain way to collect for RBC tests, that the phlebotomist inserts the needle, then waits about 20-30 seconds before drawing back to collect the blood so as not to disturb the red blood cells at the site, falsely increasing the RBC results. Do you know if this is true? I am about to order my blood tests and I want to get these (RBC magnesium and RBC potassium) right. Thank you!
Hi Lisa,
It is true that sometimes an incorrect phlebotomy technique may lead to erroneous results. A classic example is excessive fist clenching which will cause skeletal muscles to increase the levels of potassium in the tube that is not a reflect of what’s happening in the body (pseudo-hyperkalemia). Other causes for this can be traumatic venipuncture due to bad phlebotomy technique (such as “digging”) or hemolysis of the sample which will destroy cells and release potassium making it elevated.
That being said, your test is going to rather probe intracellular (i.e not plasma) potassium as well as magnesium. The only issue that I can think of here is that if your blood sample is hemolyzed (RBC broke up and the plasma will be red-ish color). Fist clenching should not be an issue here if it is not excessive. On the other hand, you can have the phlebotomist draw 1 or 2 other tubes that are not affected by venipuncture or clenching (i.e. genetic test) and then include your RBC Mag-Potassium tube next. Overall, I do not think that the 20-30 second hold time is necessary – as long as you don’t have obvious hemolysis.
Amazing we are in a chaotic dervish of one step forward, a dozen steps backwards. Make that a dozen drunken and tipsy and trippy steps backwards. Not only do we have plastizers as on culprit in disrupting the immune and endrocrine system, we have what are forever and everywhere chemicals. EPA and FDA are continuing criminal organizations, much more than some aspect of what many call regulatory capture.
So, the constellation of chemicals in our body, in the womb, as we develop, and then what the body was meant to do, and then all the resulting diseases and immune breakdowns, etc., I wonder what role those play in all the vitamin and mineral deficiancies we are seeing.
Here, one family of chemicals, killing us slowly: “The EPA announced that it planned to grant the group’s request and issued a proposed rule that would add DINP to the toxics inventory. Once the rule was finalized, companies would have to report their DINP emissions to the public database, and communities living nearby would know how much of the chemical was being released into their surroundings. In the federal register, the agency noted the science driving its decision: ‘The toxicity data clearly indicates that DINP is known to cause or can reasonably be anticipated to cause cancer and other serious or irreversible chronic liver, kidney, and developmental toxicity in humans.’
Yet more than 20 years later, the EPA has yet to make good on its promise to add DINP to the list of chemicals. It never finalized the rule, and in the intervening years, companies have continued to churn out DINP and other chemicals in its class in astounding amounts without disclosing how much individual plants make and emit. Between 2012 and 2015, as much as 500 million pounds of DINP was made or imported each year, according to the most recent numbers available from the EPA. Companies add DINP to hundreds of products in place of another phthalate called DEHP that is being phased out because it causes cancer, birth defects, and reproductive difficulties. Over the last decade, blood levels of chemicals the body forms as it breaks down DINP have climbed in the U.S., while those of DEHP have gone down.”
https://theintercept.com/2021/12/07/epa-dinp-phthalates-child-sexual-development-cancer-birth-defects/?utm_medium=email&utm_source=The%20Intercept%20Newsletter
+–+
When he was in prison in Germany after returning in 1939 from Union Theological Seminary in NYC to oppose Hitler, the German theologian Dietrich Bonhoeffer wrote the following from his prison cell before he was executed:
“Against stupidity we have no defense. Neither protests nor force can touch it. Reasoning is of no use. Facts that contradict personal prejudices can simply be disbelieved — indeed, the fool can counter by criticizing them, and if they are undeniable, they can just be pushed aside as trivial exceptions. So the fool, as distinct from the scoundrel, is completely self-satisfied. In fact, they can easily become dangerous, as it does not take much to make them aggressive. For that reason, greater caution is called for than with a malicious one. Never again will we try to persuade the stupid person with reasons, for it is senseless and dangerous.”
By stupid he did not mean that such people lacked intellectual ability, for they were often very smart, but that they had fallen under the spell of public power and lost all independence of mind. Thus he adds, “He is under a spell, blinded, misused, and abused in his very being. Having thus become a mindless tool, the stupid person will also be capable of any evil and at the same time incapable of seeing that it is evil.”
So many mindless/stupid tools in corporations, inside government, in agencies, in nonprofits, in educational settings, in fiance, in spy agencies, military. Yet, common sense hits many of us between the eyes and we know there are colluding forces that have made the human biome and our terrain rotting ghosts of what they should be.
Now, there are ways to attempt to stop the vaccine injuries in the tens of millions now on the horizon — selenium, zinc, pectin, vitamin c. Dosing is vital, and there are plenty of studies on what these 4 do to help with the toxicity of the mRNA vaccines (sic).
Lots of information on how to expel some of the immune comprimising ingredients of the mRNA vaccine (sic) — correct dosing of pectin, Vitamin C, selenium, zinc. Yet, we are blinded by scientism and mechanistic thinking.
But again, magnesium deficiency, yes, but the full terrain of the human biome is compromised by a suite of modern poisons, degrading the immune system nanosecond by nanoparticle. And if this headline doesn’t run shivers down your back, then I don’t know what would: “EPA Withheld Reports of Substantial Risk Posed by 1,240 Chemicals”
https://theintercept.com/2021/11/01/epa-toxic-chemicals-reports-withheld/
It is very useful…I need your permission to cite the theory in your paper (if you do not mind)
Hi John,
Thanks for your comment. Absolutely, feel free to cite what you need.
Dr. Lonsdale, thank you for your comment and I fully agree with you on this. I will be reading your article in Medical Hypotheses regarding these two factors. The XMEN genetic deficiency discussed here actually seems to be a good research topic regarding the important point which you raise (magnesium and thiamine). Gene knockout or knockdown can probably be replicated in animals or cell culture, to determine how thiamine-dependent pathways are affected in this syndrome. I am almost certain, because of the interplay between magnesium and thiamine, that multiple thiamine-related cellular pathways are in all likelihood going to be dysfunctional – and hence the effects on immunity seen in these XMEN patients.
This is a comment based on Dr Bilal’s post. I agree with his descriptions of the role of magnesium, but it is, in my view only half the story. The other half is thiamine because these two vital substances are cofactors to all the enzymes that have a functional part to play in energy production. The host of different symptoms from the deficiency of just these two nutrients is because they are the quintessential drivers of energy production. Therefore multiple diseases stem from the severity of their deficiency and their distribution. That is why I have written a paper in Medical Hypotheses that their combination may well be “key to disease”.