Thiamin(e), vitamin B1, is spelled with and without an ‘e’. Originally thought to be an amine, the ‘e’ was dropped when the formula became known, but the spelling using the ‘e’ is still used in many texts and across the internet. We spell it with the ‘e’ on this site because of the enhanced search characteristics e.g. thiamine ranks higher than thiamin on search engines. In addition to the discrepancies in spelling, there is quite a bit of confusion surrounding this vitamin and its derivatives used in supplements. Even the most astute readers will find navigating the world of thiamine supplements confusing. For that reason, this post will address some of the more important issues concerning these supplements.
Thiamine Chemistry
In order to understand the writing that follows, I must try to show this formula.
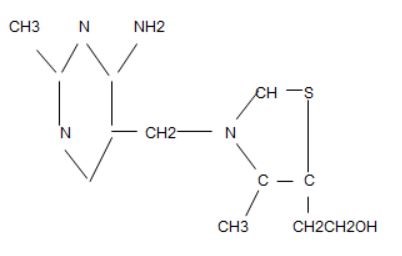
Please excuse this presentation of the thiamine formula. It was made from the Apache Open Office Drawing file. Its representation is incomplete because it does not show the “double bonds”, but it illustrates that the atoms that bind together to form thiamine are in “two rings”. The 6-sided ring on the left is called a pyrimidine ring and the 5-sided one on the right is called a thiazole ring. The CH2 that joins them is called a methylene bridge. This is the naturally occurring thiamine that we must obtain from our diet. Its deficiency causes the classical disease known as beriberi. It is important to understand the atomic construction of thiamine in the discussion that follows concerning its derivatives.
Allithiamine
The Vitamin B Research Committee of Japan, a group of university-based researchers, set out to study beriberi in detail, trying to find the best method of treatment for this disease which had been a scourge in Japan for thousands of years. Without covering the specific details, they found that thiamine was converted to a disulfide derivative by an enzyme found in garlic. Because this occurred in other members of the allium species of plants, they called it allithiamine. Thinking at first that thiamine had lost its biologic activity, when tested in animals the new compound was found to have a greater biologic activity than the original thiamine. It was found that the thiazole ring had been opened, creating a disulfide. They began a research program to synthesize a whole group of thiamine disulfides, two of which are shown below.
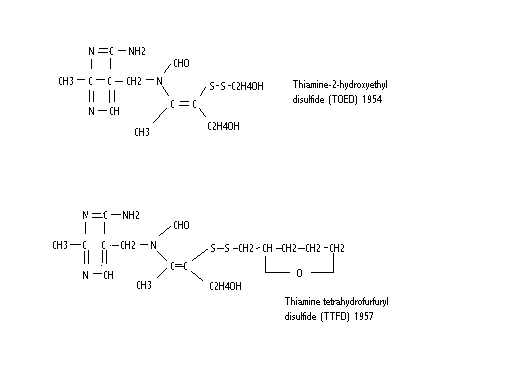
Although the arrangement of the atoms is different from the thiamine diagram, the important thing to notice is that the thiazole ring (right side) has been opened, creating a disulfide, including what is known as a prosthetic attachment (the part attached to the disulfide). A disulfide is easily reduced (S-S becomes SH) when the molecule comes into contact with the cell membrane. The result is that the prosthetic group is removed and left outside the cell. The remainder of the molecule passes through the cell membrane into the cell. The thiazole ring closes to provide an intact thiamine molecule in the cell. It is inside the cell where thiamine has its activity and so this is an important method of delivering it to where it is needed. It is this ability to pass through the lipid barrier of the cell membrane that has caused allithiamine to be called fat-soluble. It only refers to this ability, however. It is soluble in water and can be given intravenously.
This “fat solubility” is extremely important because dietary thiamine has to be attached to a genetically determined protein, known as a transporter, to gain entry to cells. There are known to be diseases where the transporter is missing. Affected individuals have thiamine deficiency that does not respond to ordinary thiamine and are usually misdiagnosed. Therefore, a disulfide derivative that does not need the transporter is a method by which thiamine can be introduced to the cell when the transporter is missing. There is no difference between allithiamine and thiamine from a biological activity standpoint. It is this ability to pass the active vitamin through the cell membrane into the cell that provides the advantage.
I performed animal and clinical studies with thiamine tetrahydrofurfuryl (TTFD) for many years and found it to be an extremely valuable therapeutic nutrient. Any disease where energy deficiency is the underlying cause may respond to TTFD, unless permanent damage has accrued. Dr. Marrs and I believe that energy deficiency applies to any naturally occurring disease, even when a gene is at fault. For example, Japanese investigators found that TTFD protected mice from cyanide and carbon tetrachloride poisoning, an effect that was not shown by ordinary thiamine (Fujiwara, M. Absorption, excretion and fatal thiamine and its derivatives in the human body. In Shimazono, N, Katsura, E, eds. Beriberi and Thiamine. (pp 120-121) Tokyo, Igaku Shoin Ltd. 1965). They exposed a segment of dog’s intestine, disconnected it from its nerve supply and found that one of the disulfide derivatives stimulated peristalsis (the wavelike movement of the intestine). It is more than likely that TTFD could be used safely in patients with post operative paralysis of the intestine (paralytic ileus).
Other Derivatives
The Japanese investigators made many disulfide derivatives, testing them individually for their biologic activity. They found that thiamin propyl disulfide gave the best results, but unfortunately gave both treated animals and human subjects a pervasive body odor of garlic. They went on to create TTFD with a deliberate attempt to remove the garlic odor and the commercial product was named Alinamin F (odorless). This is by far the best of the disulfide derivatives. Besides the trade name of Alinamin, the Japanese product, TTFD is sold as Lipothiamine in the United States.
S-acyl derivatives
The Japanese investigators synthesized a whole series of thiamine derivatives where the prosthetic group was attached to the carbon atom (bottom right C on the thiazole ring). They are all so-called open ring derivatives but the prosthetic group has to be separated by an enzyme in the body for the thiazole ring to close. The best known of these is known as Benfotiamine and several papers have been published concerning its benefits in the treatment of neuropathy. It has also been published that it does not cross into the brain, whereas TTFD does and this seems to be the major difference between Benfotiamine and Lipothiamine. Benfotiamine, a synthetic S-acyl thiamine derivative, has different mechanisms of action and a different pharmacological profile than lipid-soluble thiamine disulfide derivatives. It is predictable that TTFD would be the best choice since it has beneficial effects both inside and outside the brain and it certainly needs to be explored and researched further as a very valuable therapeutic agent.
Thiamine Salts
Thiamine is found in health food stores as thiamine hydrochloride and thiamine mononitrate. These are known as “salts” of thiamine. Like dietary thiamine, they require a protein transporter to get the vitamin into the cell. Their absorption used to be thought to be extremely limited, but megadoses are effective in some situations. The absorption of salts is therefore inferior to that of the thiamine derivatives discussed above. They are all so-called “open ring (thiazole)” forms of thiamine and represent the most useful way of getting big doses of thiamine into the cell. The reader should be aware that when we talk about big doses of a vitamin, it is being used as a drug. Although they can be used for simple vitamin deficiency, their medical use goes far beyond that because they can be effective sometimes when thiamine absorption is genetically compromised.
We Need Your Help
More people than ever are reading Hormones Matter, a testament to the need for independent voices in health and medicine. We are not funded and accept limited advertising. Unlike many health sites, we don’t force you to purchase a subscription. We believe health information should be open to all. If you read Hormones Matter, like it, please help support it. Contribute now.












It was recommended by my Dr. to try Thiamax TTFD and do 1/4 increments to test tolerance. I react oddly to almost everything so I have to be cautious in adding new supplements. I have a sulfa reaction to medications and when I opened the capsule to do 1/4 dose I had an immediate reaction to just the smell. I started coughing, my eyes burned and my nose felt itchy and burning. Needless to say, I didn’t take it and recently connected it possibly having a connection to my sula issues. My bloodwork from both a cellular specialist and my Functional Medical Dr. both agree I need this supplement, but are there alternatives to get what I need without a reaction?
You might consider a straight thiamine hcl, while less potent than a TTFD, more easily tolerated and titrated up. You might also consider rather than capsule that has added ingredients that may be problematic, purchasing it in bulk in the powder form. There is a company called Bulk supplements that carries it. I believe also a company called Nutricoast carries the powder thiamine hcl.
I apologize for including a link, but I believe it will help clarify my question. I recently came across information about Thiamine-SOLOpharm (Thiamine) and its uses, side effects, and interactions. Given the complexities surrounding thiamine supplementation and its effects on conditions like POTS and chronic fatigue, I was wondering if you could provide insights on how this specific formulation compares to other thiamine supplements, such as allithiamine or lipothiamine? You can find more details about Thiamine-SOLOpharm here: https://pillintrip.com/medicine/thiamine-solopharm-thiamine. Thank you for your help!
Good day, Dr. Marrs,
I’ve been taking 100mg of thiamax and I am not sleeping well. I don’t get a good night’s rest. What could be causing this? Should I reduce the dosage?
Are you getting plenty of magnesium and potassium as well?
Good day Dr Mars, Dr Elliot and everyone.RIP Dr Lonsdale .I’m a 23 year old girl from South Africa and I thank God for letting me come across Elliot’s Chanel which made me aware that what I’ve been experiencing might be B1 deficiency. I want to tell my story and hopefully can get advice, because I am so frustrated and demotivated .
As long as I can remember from childhood , I’d have constipation. I grew up with it and it was just ignored. Around 17years I experienced tachycardia and I saw a cardiologist who just prescribe a bcomplex and said it could be pots and I’ll just have to live with it. After many months of taking it, I stopped and the symptoms came back a year later but worse. I have IBS characterised by chronic constipation and I would still experience low energy, fatigue , palpitations , cold hands and feet, terrible circulation , would wake up suddenly at night scared, panick attacks and anxiety. After I’ve discovered thiamine deficiency I looked for all thiamine and but can only find Thiamine HCL in my country. I take 100mg a day with magnesium glycinate along with nutritional yeast and I have seen little improvement especially with anxiety and panick attacks but I still experience pots symptoms sometimes. My diet is bad as I would eat rice every day but I’m trying to cut out sugars .
My question is it possible to one day function without needing thiamine ? Was there anyone who took any form of thiamine and and was cured ? Or will it be forever? Do you think if I change my diet and supplement thiamine for a long time , I’ll be healed? What type of diet is recommended? Keto? Carnivore? What general advice can you give me?
I’m sorry for the many questions , I am just sick and tired. I’d greatly appreciate your advice.
With thiamine hcl you may need more than 100mg. It’s not as well absorbed as the other forms. Consider increasing your dose gradually to find a dose where you begin to see improvement. Increase and hold a week or two and then repeat. As far as improving, yes absolutely. It is likely you may need additional vitamins too. Consider a good multivitamin. Finally, yes, diet is important. What form of diet you ultimately decide to use, the bottom line is to cut out garbage processed foods. Whatever you eat should be as nutritious as possible. You want to add nutrients to your diet, not toxins like those found in processed foods. Good luck.
Thank you very much for your fast response .
In 2019 our daughter was given 2000mg metronidazole as a single dose and the side effects from it were horrible. Not through doctors, who just treated anxiety symptoms, but through Erin Jensen’s blog and Hormone Matters we realized it caused thiamine deficiency and started thiamine hcl along with magnesium l-threonate. Titrated up to 600mg thiamine hcl daily over 5 weeks and all symptoms ceased. This lasted 4 1/2 years until late March of this year, 2024, she gave plasma and had a total relapse. She had been taking 400mg thiamine hcl for past 6 months so we added thiamine hcl 100mg at a time but found no relief. Switched over to lipothiamine 50mg and got up to 150mg lipothiamine a day. Tried 100mg in a sigle dose and had horrendous paradox which gave her such a panic attack we took her to e.r. They did mri and ct which showed no problems in brain. Just being awake seems to calm worst anxiety along with talking to others. We came home and took a few days of 500mg hcl a day and one lipothiame 50mg. Today starting 50mg lipothiamine in morning and 50mg lipothiamine in evening with a hcl at 4 p.m. and 4 a.m. if needed. Taking one capsule magnesium l-threonate twice a day. Daughter has great anxiety, just after waking up is the worst with stuttering and feeling very afraid and very panicky. Most of the wobbly legs is gone. Finally get to see a functional doctor Tuesday. Doctors at hospital were not listening when we though B1 infusion might help. They saw her B12 showed high on bloodwork and said she had plenty of B vitamins in system so they wouldn’t try the B1 i.v. No matter what, we will continue the lipothiamine and slowly raise the amount orally. Thank you for your information and all your teaching or our daughter would be totally lost.
It may be worth taking a break from the thiamin for a week or two and then starting back at a lower dose. People that take B1 for Parkinsons have found that to be helpful when their symptoms start to worsen again. You may be familiar with this but there is a gentleman that is a nutritionist that has done a lot of work researching B1 and he has a YouTube channel called EO Nutrition. You may find some of his information helpful as well.
Greeting Dr. Lonsdale,
Is it possible/safe to nebulize a powdered form of thiamine such as thiamine hcl or thiamine mononitrate (or some other form)?
Thanks for all of your hard work and dedication,
Brian
It is possible!
Here’s a video where I demonstrate nebulizing 1/16 tsp (=100mg) of powdered Thiamine HCL dissolved in distilled water with no ill effects:
https://rumble.com/v406rv7-can-you-safely-nebulize-vitamin-b1-thiamine.html
Per Dr. Antonio Costantini (https://highdosethiamine.org/hdt-therapy/), 100mg injectable thiamine = 14000mg oral. There are some who believe that nebulizing a medication is equivalent in potency to injecting it because the GI tract is bypassed in the process.
Have fun!
I started getting peripheral neuropathy in the middle of September 2023 which responded incredibly well to benfotiamine 250mg, which I now take 3 times a day. It initially improved my sleep and took away most of my symptoms of peripheral neuropathy so that most of the day I could forget I have it. I am also taking a methyl B-complex, magnesium threonate and bisglycinate plus electrolytes in the form of coconut water. But over the past week I now can’t sleep. I feel hot-wired. I’ve tried reducing the benfotiamine, particularly the evening dose but my symptoms of neuropathy reappear big time. I have also ordered some R-alpha lipoid acid and acetyl-L-carnitine. Do you have any advice.
According to the article up above benfotiamine DOESN’T cross the BBB (Blood Brain Barrier) whereas TTFD (Lipothiamine) does. So perhaps try switching to TTFD (Lipothiamine) and see if your sleep improves after awhile. God bless!! 🙂
250 x 3 a day of fat soluble benfo cannot be safe long term. Way too high and unnatural, the body has no idea what to do with it.
Have had good results with recovery from CFS utilising 1500mg of thiamine hcl a day, what would be a equivalent dose using Benfotiamine
Does this mean that we can increase the absorption of thiamine mononitrate or hcl by eating it with garlic? Would this be meaningful?
Hello Dr Lonsdale and Dr Marrs,
Thank you for all of your support in this area!
Thiamine injections and IV infusions:
My naturopath can do these for me – can you recommend the form and dosing for injections or IV?
Liquid B complex:
I have ordered allthiamine and found a liquid b complex that I am taking and it doesn’t make me feel nauteous unlike the other b complex capsules I have taken. Found on amazon “liquid b complex solution”
Magnesium:
I am taking magnesium citrate as I find this very compatible is this a good form to take along with the other b supplements?
In deep gratitude for your work,
Kate
Dr. Lonsdale, thank you for all your work. I recently read your book on thiamine deficiency and your contributions on Hormones Matter.
I have a question regarding maintenance dosing after thiamine deficiency has been addressed: Is 50 mg Lipothiamine long-term overkill and/or would a combination of different forms be helpful?
For context: I was able to build up to 2000 mg thiamine per day (500 mg HCl, 500 mg TTFD/allithiamine, 1000 mg benfotiamine) and kept the dose for two months. Now I try to lower it to a more sustainable long-term dose. I have several SNPs of which some might be relevant for thiamine transport incl. SLC19a1, MTHFD1, MTHFR, MTRR.
thiamine is non-toxic. Use the dose that works for you
Thank you for the quick reply!
Two follow-ups:
– From the published research – mainly from Japan and China – TTFD maintenance doses range from 50-100 mg. Does this dosing seem reasonable?
– Do you have a sense or is there published data on the difference in absorption/ hydrolysis of TTFD of regular capsule vs. enteric coating? You mentioned in a previous comment that coating helps “to avoid some loss of the intact molecule by hydrolysis from stomach acid”. I was wondering how big this effect is.
Hi Dr Lonsdale,
I have been suffering from vaccine injury over 10 years and have extreme chronic fatigue, dysautonomia, neuropathy, POTS, tachycardia, and a list of more symptoms… I started on Allithiamine almost 2 years ago and have some improvements. When I was using regular thiamine I was still extremely deficient in thiamine, after taking Allithiamine my levels finally came up. Now I found out I am pregnant. Is Allithiamine safe while pregnant? I was on 100mg Allithiamine per day before finding out I was pregnant and need to know what dosage to take while pregnant please. And is there a way to get an appointment with you?
read the posts on this site
Thank you I have seen it is safe just wanted to make sure the sulfide part of it was ok for the baby. Do you know women that have taken Allithiamine while pregnant? What is a good dosage? Is 100mg too much?
Wow, you are curt
Thank you for this great info!
Can you please clarify the differences among allithiamine, lipothiamine, Thiamax, HCI, Sulbutiamine, and Benfotiamine?
I am confused on which should be taken and by whom…
Many thanks in advance!
If a person believes they are thiamine deficient is doing B1 injections (sub q)–THIAMINE HYDROCHLORIDE USP (Sandoz or Cytex) a good option? Or better to stay with pills?
What do you think about Liposomal Tiamine? In that form it goes into cells directly and it’s a long lasting form.
Hello,
Would taking Benfotiamine (or even thiamine HCl) alongside DMSO improve its absorption to get into the cell and cross into the brain? Could it then come closer to the effectiveness of TTFD?
Thank you Doctor
No
Could you please explain why dmso could not be used to help take B1 more efficiently to destination?
No he cannot reply more then a few words.
Hi
Can our body have problem with converting thiamine into its active form?
Yes
Would you please explain a little, is it related to mthfr mutation? And should we go for foods instead of supplements like thiamin hcl?
Thank you
Thiamine in the diet is inactive. It has to be activated by attaching 3 phosphate molecules to create thiamine monophosphate,diphosphate and triphosphate
Dr Lonsdale or Dr Marrs,
Would using a Coenzymated version (one that has the phosphate molecules attached) overcome the conversion issues then? Also would such a version still cross the BBB and get into the cells so that it could be used as a drug, as you mentioned, for correction of energy deficient disease states?
Thank you for your time.
Yes. Source Naturals makes the coenzymated form. Sublingual.
Hello,
My daughter (as ell as my other 2 children to a lesser degree), has been suffering for years with health problems,and no one can offer answers or relief to any of her symptoms. Migraines, anemia, muscle pain, joint pain, POTS type symptoms, fatigue, muscle spasms, Abdominal pain, urinary problems, mood/anxiety problems, cognitive decline and brain fog, vision issues and I am sure I am forgetting some. I found you research and ordered benfotamine before I read about TTFD. Is it ok to start her on the benfotamine until I can get some TTFD? She is 17 and weighs aroudn 130 lbs, what dosages would you recommend and type of magnesium etc. I live in Ohio, I am not sure if you are still practicing, but I would love to bring my daughter to you in person, or perhap an apprentice of yours?
Thank you so much for your help!
Hello
I had a lot of those symptoms and it was all connected to Dairy intolerance and food and drinks that had Lactic and citric acids in which is in processed foods and drinks, I can’t have fermented things either, so I follow a low histamine diet without Dairy and dairy starter cultures and all my symptoms went away, AMAZINGand life changing. I hope your daughter finds the answers to her health problems.
Take care
Jackie
Most of the symptoms mentioned by Jackie can be created by a problem with oxalates-the absorption of and the body’s ability to utilize oxalates. You can develop problems after changing your diel drastically or taking antibiotics. They are found in all foods except meats. Sally Norton is one of the leading authorities on oxalates has a book and also utube videos available. We had minor symptoms of oxalate issues but didn’t know it was actually affecting us because we had never heard about oxalates except the role they played in kidney stones. It hit us hard when we changed our diet from standard American to Keto and then went back to the standard American diet. Some of our symptoms bad cramps, sharp pains in different parts of our body, gritty eyes, blurred vision, urine looks foamy and sandy. brain fog, severe itching, bladder infections. Some of the foods that give a bad reaction: almonds, chocolate, tea, spinach, cinnamon, wheat, potatoes, broccoli
All of these symptoms can also be cause by taking an antibiotic in the fluoroquinolone class of drugs. Google ‘floxed’ and you will see. https://fq100.org/
I don’t understand why an article titled Navigating Thiamine Supplements doesn’t mention most of the various forms of thiamine that consumers are faced with when consumers go shopping.
I see that some of the above comments talk about these various forms, but Mr. Lonsdale what are YOUR opinions on the differences between coenzymated, TTFD, Allithiamine, benfotiamine, and ordinary thiamine HCL?
Read the post again. The details are there
Dear, Dr. Lonsdale:
In searching Google for “cocarboxylase chloride vs ttfd,” I found the above article. I just wanted to thank you for your writing and this website — which I plan to come back and visit again soon.
May God bless you with health, happiness, and prosperity.
Best wishes,
Aaron
Dr. Lonsdale & Dr. Marrs, I was diagnosed with Parkinson’s last year, Symptoms began in 2018. I and 3 of my 4 siblings plus my mother are hypothyroid. I have been taking levothyroxine for over 10 years. I began taking thiamine HCL following Dr. Constantini’s protocol at the end of July 2021 hoping to improve my PD.
I was surprised when I saw my primary doctor. My THS for the last 3 years on 88mg Levothyroxine was, 2.55, 1.49, 1.69, 1.03, 1.80. After the Thiamine my results came back at 0.06. After reducing and still getting 0.28 I am now trying no Levothyroxine. WOW! took me by surprise. After that I found your site and read your book. My questions are does it seen possibly genetic that myself and family are not absorbing B1 properly? Might I be better off trying TTFD? I have not yet observed an improvement with my PD on Thiamine HCI, 3G split into 2 doses per day. Also if I try TTFD should I stay away from Lipothiamine? I believe I read that ALA is not good for the thyroid. And finally can you offer some point of reference as to how much TTFD I should start at compared to the Thiamine HCI. For reference I ma 5’8″ tall and 142Lb. I try to eat good, ex: drink water not sugary drinks, try to eliminate most added sugars, lots of fruit and vegy’s etc. Thank you for your time and thank you for your work.
My daughter has no ankle or knee jerk present. Her other obvious symptoms are brain fog and constipation. She is 12 years old. Would she benefit more from Benfotiamine or Allithiamine (or thiamine hcl)?
Probably any of them
Hello,
My daughter has red patches in her throat, slightly elevated Eosinophils and basophils, slightly elevated A1C after fasting. She is Asian and adopted so I don’t know genetic history. She has been eating more carbs and white rice over the last several months than we used to eat. I came across your case study about sugar sensitive Esophagitis. I noticed a slight stiffness in one knee this evening. After your article I gave her brewers yeast as I had that in the house. Is that adequate? I am also concerned these symptoms could be related to parasite infection due to her early living conditions. Have you seen a connection there?
I’ve been on 50mg allithiamine for about 2.5 weeks and feel horrible but trying to ride it out. I have some magnesium on the way.
The last few days I’ve had a weird taste that’s not related to anything I’ve had to eat. It’s not garlic, but definitely like a seasoning. Has this been observed elsewhere?
I had a very bad reaction to a sulfa drug when I was a kid. From my Googling I can’t figure out if this is a potential problem with taking thiamine or not. Any info would be appreciated.
Read about paradox on this forum
Is vitacost’s version of B1 acceptable? My dad uses it in conjunction with c/l. Would you suggest another 500 mg thiamine hcl supplement in place of that one? Thank you sir.
I have just today found this site and read for hours the thiamine deficiency connection with dysautonomia. Thank you for your many years of sincere and serious medical inquiry and devotion.
My husband is an internal medicine physician of 25 years and, like many (most) in Western medicine, seems to poo poo the idea of vitamin/mineral deficiencies. I am now 50 and experiencing perimenopause and all the lovely changes that accompany (sarcasm) while homeschool our elementary aged children, so I am accustomed to stress. Until 8 weeks ago I was in very good health and spirits. Solid and stable Body weight and fitness levels. Cardio and resistance Exercise several times per week, primarily meat/veggie/fruit diet and no medications other than Mg, D3/K2, NAC, quercetin, probiotic and fish oil. Biweekly acupuncture sessions for hormonal reasons.
Ten weeks ago I developed a Paronychia from gardening without proper gloves and had to take doxycycline to clear the infection. That same week I had to euthanize our beloved boxer dog. Of course much sadness and grief followed. Within 10 days I contracted Influenza A for the first time in 25 years. While the fever lasted only 48 hours the weeks that have followed have been a nightmare come true. Extreme fatigue, tachycardia RHR >90, dizziness, lack of appetite, shakiness in arms and hands, weakness in legs and last week a diagnosis of dysautonomia-cardio/neurogenic syncope. Full cardio workup and many blood panels all normal with exception of low iron. To go from feeling 100% everyday to being terrified each morning of what the day brings is life altering not only for me but our children. I had begun taking a B complex from Pure Encapsulations and will continue while waiting for your recommended TTFD to arrive in the delayed holiday mail.
Finally, my question….should I continue to take the B complex for several days to a few weeks, titrate or switch over to the TTFD?
Thank you in advance for your time and expertise.
Sincerely,
Dawn Vargas
From Dr. Lonsdale:
This is a typical and repetitive story. Tell your husband to remember that the body is a biochemical machine and if we don’t know how it works, we are like car mechanics who don’t know anything about the car engine. In the seventies I was a pediatrician at Cleveland Clinic and I saw many children with ADD, hyperactivity etc. Their diet was appalling. Full of empty sugar calories, so I studied sugar metabolism and became aware of its complexity. I found that thiamine was the most essential factor, so I gave them thiamine and they became normal kids. O, I paid for it. I became persona non gratis and resigned. I became an Alternative Medicine doctor and I have been enrolled in the Hall of Fame of the Orthomolecular Society in Toronto, founded by Linus Pauling. My hard won experience has taught me that ALL disease is due to energy deficiency.. Tell your husband to go on line to Hormones Matter. I have written many posts on it and you will learn how to treat your symptoms. Now that an Italian doctor has published his cure of Parkinson’s disease with megadoses of thiamine, I believe that nutrient based medicine is indeed the medicine of the future. Tell. your husband to get our book on Amazon books, Lonsdale D, Marrs C. Thiamine deficiency disease, dysautonomia and high calorie malnutrition published by Elsevier in 2017. I have lots of publications in the medical literature. It has long been my opinion that merely treating symptoms is defrauding the patient.
Thank you for the quick reply. I will order the book for my husband. He’s slowly come around to the benefits of acupuncture. A physician in his group left this year to practice functional medicine. I will be switching to an integrative MD soon.
To my specific pressing question? Shall I keep taking the B complex currently taking from Pure Encapsulations or switch to the Lipothiamine once it arrives in the mail?
Thank you, kindly,
Dawn
Thank you for the quick reply. I will order the book for my husband. He’s slowly come around to the benefits of acupuncture. A physician in his group left this year to practice functional medicine. I will be switching to an integrative MD soon.
A b complex or good multi-vitamin is often needed with thiamine. Once thiamine is on board, other deficiencies may present themselves and so it is a good idea to take a multi. Also, be aware that additional electrolytes will likely also be needed. Magnesium will be important as will potassium, and sometimes calcium, salt and phosphate depending upon diet and other factors. Tracking your diet with a program called Chronometer will help pinpoint where you might have vitamin or mineral deficiencies. If you take medications, many deplete different vitamins and minerals and so do a google search by medication to see what else you might need.
Great! Thanks for the info. I take a womens multi daily and use LMNT hydration solution in the 2-3 liters of water I drink daily. Again, appreciate the time and expertise.
Thank you for the suggestion of Chronometer. Will check it out.
I feel confident I get appropriate macros each day however, kI am less confident of the micros. I did have a Mg deficiency 5-6 years ago that was quickly remedied with supplementation. I pray the current symptoms can be as easily relieved with TFFD.
I eat a pretty basic diet of lean animal protein, vegetables and fruits, olive and coconut oil, with occasional sprouted grain and cheddar cheese, 2-3 liters of water per day with LMNT electrolytes.
I do not currently take any prescription medications although the MD’s want me to take Toprol XL and Lexapro and Progesterone cream to manage the symptoms of dysautonomia and perimenopause. I do take a large quantity of supplements. Perhaps one of the below affects thiamine absorption?
Women’s Over 50 Multivitamin
Mg Glycinate (switching to taurate per this site)
B Complex Plus
NAC
Quercetin
Vit C
Vit D3K2
Probiotic
Fish Oil
Iron for low ferritin levels during this time too
I am hopeful the thiamine TFFD supplement will resolve the unnerving symptoms I have had for over 2 months post flu infection. Until this time period I have always been active and fit with loads of energy. I cannot fathom the thought of walking this path for years as have many of those in the comments section have.
Again, thank you kindly for your wonderfully informative website and for being so generous with your expertise and time.
May God bless you and keep you,
Dawn
Sunflower seeds, sesame seeds and whole grains are foods highest in thiamine. Use the tools on my food data
Seeing good improvements so far with the B1 Thiamine HCL 100mg and a B complex with Magnesium Taurate. Wondering if there will be any further benefit of increasing the thiamine HCL to 200 mg (for a total of 300 mg including what is in the B complex) or switching to allithiamine?
Thank you in advance
Dawn
I’ve been suffering for 2 years now with a condition doctors can’t find an answer for.
I used to eat a diet heavy in sugars and carbs and then noticed numbness forming in my fingers and toes. It took months to learn that it flares up whenever I consume any amount of sugar and even when I eat salt.
My mother’s cancer doctor mentioned that it sounded like I had a major b12 deficiency and that I should try supplements. I did and it worked for a time, giving me improvements, but there was still residual numbness. Which gradually got worse.
I took more and more b12 just to maintain myself, trying loads of supplements in the hopes of some of them helping. When I got blood testing, they couldn’t find anything. They checked for deficiencies and didn’t see anything.
But the b12 was the only thing that made me feel any better, even though the constant state of apathy I lived in. I had no energy and no strength whatsoever.
I took massive amounts of b12 supplements and ate beef liver and salmon everyday. The beef liver was the major thing that would make me feel okay, and I didn’t understand why. I’d tried b1 supplements before, but these were all the water soluble form.
Well, I later learned that it’s carbs that trigger me too, 2 years in. Since quitting carbs, I’ve regained an incredible amount of feeling in my body, though this leaves my food choices quite limited.
Even eating a single carb will trigger numbness, brainfog and pain that’ll set my healing back for days. Sugar is worse and the feeling doesn’t really return if I eat any of that. Not to mention the depression is causes me. Salt is the least destructive and “only” leads to me ending up feeling numb and sad for some hours. It’s greatly influenced by how much I take in and how often I eat it.
I started taking allithamine and got wondrous results. Faster healing, more feeling than I’ve had in ages, and my mood has improved. Has this all really just been a b1 deficiency the whole time?
I’d have never found this normally, given how rare fat soluble b1 is. I had to order this stuff from amazon. And I tried loads of benfotamine, but it didn’t really help beyond improving my mood. I had some body pain from taking it and stopped, not noting a big difference.
Well, maybe the issue was that I didn’t eat enough fat when I took it? I eat 2 salmon fillets whenever I take the allithamine and it works great to help me absorb it.
The strange thing is that my body reacts even to artificial sweeteners and stuff like alluose, which your body doesn’t even really absorb. It makes me question if there’s something wrong in my bowels and there’s bad bacteria feeding on stuff I put down there.
But I spent a lot on anti SIBO pills and did a huge regiment of them, along with taking probiotics and prebiotics.
But maybe eating lots of inulin wasn’t the best idea? It’s got carbs, so perhaps the bacteria fed off it too?
I’m wondering if you’ve ever seen anyone who had symptoms like mine and if this can all just be chalked up to a b1 deficiency. That maybe I’ll be able to eat something other than meat in the future.
I miss being able to eat normal food but it causes me so much misery and grief that I can’t.
You have vitamin B1 deficiency, not B12. Stop taking carbohydrate junk and start eating God made (organic) food. Read the many posts on Hormones Matter and educate yourself.
Make sure that the B12 is in the form of methylcobalamin, not cyanocobalamin. The good gut bacteria need B12, but cannot convert the synthetic form, cyanocobalamin, into the natural and active form, methylcobalamin. Only the liver can make this conversion.
Correct, nobody should be taking cyanocobalamin. But methylcobalamin is not the ideal default alternative. Overmethylators, or those with significant methylation imbalance/dysfunction, should not jump to methylcobalamin. Negative side effects can occur due to further imbalance in methylation.
A safer and often more effective default alternative is either adenosylcobalamin or hydroxocabalamin. Hope this helps people that are reading thru these comments.
Is there a specific type of thiamine for those of us who have CBS mutation? Like most of us my issues span decades and I won’t go into all the diagnosis as it just complicates things. I no longer have insurance or the ability to work and insurance doesn’t cover most of the doctors who actually help instead of hurt. I have been unable to tolerate sulphur donors due to my mutation (amongst other things). I’ve been trying different types of moly to no avail.
I’m doing my best to do all my own research and read through the comments in case it’s already been asked.
Hi Derrick.
Just want to say thank you for all that your write, it is very helpful!
What are your thoughts on sulbutiamine? I have tried Allithiamine (TTFD) but don’t seem to get a great response from it. Is sulbutiamine considered an effective form?
I also seem to get more benefit from befotiamine than Allithiamine; the difference is very noticeable though I did not take for a long period of time.
Thanks a lot
Daniel
Hello Dr,
My 9 year old has autism with speech delays and speech clarity issues and and while he responds well to thiamine and magnesium (Along with magnesium) the gains are short lived. He has slc19a1 and slc19a2 mutation. Low neutrophils, high lymphocytes, low mcv, low mch and low heamoglobin. What would be the best form of thiamine? Is thiamine Hcl okay to prevent hearing loss?
I treated 10 autistic children with TTFD (Lipothiamine) in a pilot study. 8 of them improved
What doses/protocol for Autism kids?
Would it be ok to just eat crushed raw garlic rather than Ttfd? If yes, how much?
Hello Dr. Lonsdale,
Is it ok to take benfotiamine and Allithiamine at the same time? I am planning on switching to Allithiamine or Thiamax but don’t want to stop benfotiamine that I currently take immediately until I am at optimal dose of TTFD.
Also, the B-complex I take all food based, is that ok to take with TTFD or do I need Thiamine HCL separately?
Yes but you don’t need to
Dr. Lonsdale, when taking thiamax i get very bad stomach cramps and get a garlic/sulfuric smell in the nose which subsides only after many hours. Is there anything to do about it?
Thank you.
Thiamax was a result of clinical research by Elliot Overton in the UK. It contains TTFD, the synthetic counterpart of allithiamine, the thiamine derivative that occurs in garlic. Thiamine propyl disulfide, an earlier derivative, had to be discarded because it gave an intolerable garlic odor from the patient. TTFD was a deliberate effort to make a disulfide derivative that did not give rise to this odor. However, I suppose that it might in some people.
Hey Doc! So if I’m hearing you correctly, Allithiamine is in it’s natural state, as would be found in garlic? Or has it been modified to put in supplement form? As some others have stated, I’m having “side-effects” from thiamine HCL even though it is clearly resolving one of my long-standing symptoms. Wondering if Allithiamine, being a more natural form, might be tolerated better. I’m also trying some nutritional yeast, as it’s high in thiamine.
Allithiamine is the molecule isolated in garlic. TTFD is the synthetic chemical version derived from studying allithiamine. TTFD, in particular, is less odorific, so more suitable as a substance that more people are comfortable ingesting.
Hi
I wonder if you can tell me if supplementing with thiamine increases histamine levels? I have POTS, histamine intolerance and suspected MCAS. im very suspicious of a thiamine deficiency due to the onset of orthostatic intolerance over 10 years ago post vaccination (gardasil but could have been another) . I dont eat many foods high in thiamine due to their inflammatory impact on the gut. (Beans, grains etc).
Thanking you in advance
Histamine is released from cells when the cells receive a signal from the brain. The signal requires energy that in turn requires thiamine. POTS is an energy deficient disease.
Thank you so much for your reply Dr Lonsdale. We will follow your advice. It is deeply appreciated.
Hi Genie. I was curious as to whether you are noticing a difference since adding the thiamine HCI? I struggle with ocd as well. I hope it is helping your son. OCD is terrible!
Dear Dr Lonsdale,
Can thiamine supplements help with Ocd? My 13 year old son suffers with anxiety and Ocd. He is on Fluoxetine 10mg daily with little success but I don’t want the dose to be increased. If thiamine would help then what dose would you recommend? His diet is good and sugar-free. He takes a multivitamin, 400mg magnesium and fish oil capsules daily. Can he take thiamine with the Fluoxetine at a reducing dose till we get him off it? I know you are a really busy man and I hope you find the time to as answer this. Any advise would be greatly appreciated. Every blessing for continued good health and God bless you and your great work.
Thiamine can help with OCD and/or anxiety, but you have to think differently. OCD and unnecessary anxiety are caused by energy deficiency in brain cells, either because of diet or a genetic reason. Taking high dose thiamine stimulates energy metabolism. Get him off the drug and start with about 1-2 100 mg thiamine HCl. Read the many posts on this forum.
Hi Dr. Lonsdale,
I tried to order Lipothiamine, but it appears they have either discontinued it or are back ordered. I have started taking Allithiamine 50mg daily, but am getting some rather unpleasant side effects from it—not sure if this is a temporary thing while my body adjusts. I also am extremely sensitive to medications and supplements (we had tried 100mg thiamine injections and after the first dose, realized it was too high for me). My movement specialist doctor has me on it due to essential tremor, but I do believe based on other symptoms I may be low in B1. I wanted to get your thoughts on this.
Also, have you heard of the supplement Thiamax? Just wondering how it compares to Allithiamine and Lipothiamine.
Read about “refeeding syndrome” on this website Continue 50 mg of Allithiamine until the “side effects” improve. Then titrate the dose up as the symptoms improve.
Hi Melissa, Thiamax is Allithiamine. Go slow and good luck!
Hi Doctor Lonsdale and Marrs,
Quick question about le other B vitamins we need to take along with B1. What dosages do you recommend? The B complexes I could find OTC vary wildly in dosage, sometimes to a factor 10. Also, is there a specific B that is more essential than another (B12 and B6 seem more important?)
Thanks!
All together they represent a team. I take 250 mg of B complex
Thanks. Any specific formula you would recommend? I find they come in such a variety of relative % it is hard to choose.
Dear Dr Lonsdale,
I am so grateful for everything I am learning from your book and this website!
One question regarding B-complex supplementation in conjunction with large doses of allithiamine:
Is there any danger of overdose or toxicity when increasing consumption of a B-complex with the following composition (contains not only B-vitmins!) above recommended daily allowance?
Per capsule:
Biotin 400mcg
Choline (Citrate) 28mg
Folate (L-5-MTHF) 667mcg DFE
Vitamin B1 (Thiamin HCI) 110mg
Vitamin B12 (Methylcobalamin) 400mcg
Vitamin B2 (Riboflavin 5′-Phosphate Sodium) 10mg
Vitamin B3 (Niacin) 10mg
Vitamin B3 (Niacinamide) 130mg
Vitamin B5 (Pantothenic Acid) 110mg
Vitamin B6 (Pyridoxal 5′-Phosphate) 10mg
There is no known toxicity from B vitamins apart from “paradox” at the doses we are recommending.
Thank you for your reply!
What dose of B-complex would you recommend together with, for example, 150 mg or 300 mg of allithiamine?
Dr. Lonsdale,
Pyridoxine hydrochloride, the synthetic form of what most supplement makers (putatively) call “vitamin B6,” would be an exception, wouldn’t it?
“Pyridoxine induced cell death in a concentration-dependent way in SHSY5Y cells. The other vitamers did not affect cell viability. […] “Moreover, both pyridoxal-5-phosphate dependent enzymes were inhibited by pyridoxine. In conclusion, the present study indicates that the neuropathy observed after taking a relatively high dose of vitamin B6 supplements is due to pyridoxine. The inactive form pyridoxine competitively inhibits the active pyridoxal-5′-phosphate. Consequently, symptoms of vitamin B6 supplementation are similar to those of vitamin B6 deficiency.”
Vrolijk, M. F., Opperhuizen, A., Jansen, E., Hageman, G. J., Bast, A., & Haenen, G. (2017). The vitamin B6 paradox: Supplementation with high concentrations of pyridoxine leads to decreased vitamin B6 function. Toxicology in vitro : an international journal published in association with BIBRA, 44, 206–212.
I hope that Dr Lonsdale can address this one..as well as the confounding relationship that seems to exist between B6 and B1. Susan Owens explains it some, but only moslyt reassures me that we know very little.
Did you ever get an answer?
“There is no known toxicity from B vitamins” not with Vitamin B6 as long term use will cause peripheral neuropathy.
Dr.Lonsdale,I am 100% convinced that I have a severe thiamine deficiency however I am having issues with supplementing it.I am having weird reaction from taking even a 10 MG pill of Thiamine HCl.Weird throat swelling and dizziness.I have tried Sulbuthiamine too same thing.Can you please tell me if this is an allergic reaction?I am really worried about taking more and no one seems to know.
Thank you!
You are suffering paradox or refeeding syndrome. Find out about it by exploring the many posts that explain it.
The throat swelling may be an allergic reaction to the excipients in the products while the dizziness is likely what Dr. Lonsdale suggested, a paradoxical reaction. I separate the two because many supplements contain additional substances for processing, storage, stabilization, etc. of the vitamin that are allergenic to some people. Even the gel cap can be allergenic. Read the ingredients and I suspect you’ll find these additional substances and any one of them can cause a problem. Going forward, find a clean version of thiamine, a brand that contains as few of these extra ingredients as possible.
Thank you both very much! I just checked, all the pills that i have contain the same additional substances.
Perhaps buy allithiamine powder, if you can find it, and, if not, try sulbutiamine powder, which is much easier to purchase online through several venders, often in the field of “nootropics” (were, presumably, it has a nootropic effect precisely because it, like TTFD, crosses the blood-brain barrier easily).
For sulbutiamine, a flat 1/64th of a teaspoon (you can buy mini-spoon sets on Amazon), or 0.076 mL, provides about 100 mg of sulbutiamine (which is typically sold in 200 mg capsules). This would allow you to not take in any excipients.
Sorry, but I don’t know how much sulbutiamine is equal to how much TTFD. I do know, however, that they are both thiamine sulfide derivatives and should, per Dr. Lonsdale, have a similar mode of action. That said, maybe you can find a source of TTFD powder that I’m unaware of.
Dear Dr. Lonsdale,
I was having some memory issues that improved to a decent extent while taking Lipothiamine. The issues may be related to a period of sustained drinking in the past with my Neurologist suspecting Wernicke’s Encephalopathy but is not entirely sure as there were no MRI indications of Wernicke’s specific findings but rather” findings of Mild generalized parenchymal loss for the patient’s age and Several scattered punctate T2 /FLAIR signal hyperintensities in the cerebral white matter -non specific”. The neurologist wasnt concerned about the white matter hyperintensities as he said they were minor but suggested that perhaps the alcohol consumption together with stress could have caused a mild case of WE based on my symptoms.( that seem to have stopped progressing.)
It’s a presumptive diagnosis at best because I didn’t have symptoms at onset that are typical of WE.
I’m wondering what to take as it seems that Lipothiamine is off the market and my understanding is that Allithiamine, since its not enterically coated wouldn’t do the job and some are saying to take it topically. (Im not even certain how one does that or the amounts needed topically )
Any clarification on the above would be greatly appreciated.
Thanks!
Michael
Dear Dr. Lonsdale,
My Neurologist has given me a presumptive diagnosis of Wernicke’s Encephalopathy. The symptoms at onset and now were/are relatively mild for WE and although he didnt find any MRI findings that are specific to Wernickes based onmy history : a recent unfortunate first time bout heavy alcohol use which it seems like I had stopped in time , my symptoms and “mild generalized parenchymal loss for patients age” reported on my MRI, felt that I had a mild case of Wernickes that seems to have stabilized/halted.
I had been taking Lipothiamine and I’m not certain if that helped with the lack of progression but my symptoms in terms of memory if not balance seem to have improved since I started taking it. (@200mg a day split in 2 doses) . I would like to repurchase it as I ran out but it doesn’t seem to be available anywhere. I was wondering if Allithiamine would be as effective even though its not enetrically coated and if not would you know if there a source of Lipothiamine or a similarly encoated ttfd supplement available ?
Thank you for your help!
Michael
Allithiamine is good and there is also a new TTFD product on the market called Thiamax from Objective Nutrients. It is available on Amazon now. It comes in 100mg capsules.
Hi Dr. Marrs,
I realize that I posted almost the same question twice.( I hadnt seen my post appear so thought it got lost somehow, now I see them both-) My apologies.
So you are saying that Allithiamine or Thiamax would work as well or almost as well taken orally as Lipothiamine?. ( I was given pause because I had read that stomach acid could degrade part or all of the non enterically coated forms of TTFD.)
If thats true, that Allithiamine or Thiamax would be as effective or almost as effective as enterically coated TTFD would that also be the case if I had some form of WE if whatever I have was caused by alcohol or if there was an alcohol history ? My understanding although I could be wrong , is that if issues have been induced by alcohol then things change in terms of treatment course and prognosis.
Would upping the dose orally on Allthiamine theoretically compensate for any alcohol induced issues?
I do know that I was feeling a lot better on the Lipothiamine and wondering whats the best course now in terms of supplementation given my situation.
Any light you could shed on this would be greatly appreciated.. Thanking you in advance for your time and help.
Best Regards,
Michael
Yes, Allithiamine and Thiamax should work just as well. You may have to compensate a bit for the lack of enteric coating and you may require a an equivalent dose of alpha lipoic acid (ALA) separately, but these two have the same TTFD formulation as Lipothiamine. The Allithiamine is made by the same company as the Lipothiamine, even though the two are listed under different names. Thiamax is made by a new company formed by Elliot Overton after reading our book and seeing a need for a higher dose TTFD formulation. I would suggest though, if you were seeing improvements on Lipothiamine, do not hesitate to get started on either the Allithiamine or Thiamax. Letting the thiamine lapse will cause a relapse in symptoms.
With regard to the ALA, only a low dose, equivalent to what you were getting in the lipothiamine or less. High dose ALA in someone who has thiamine issues causes all sorts of problems including leaching thiamine. It’s a tight balance.
Many years ago, I suggested to the vendor that perhaps a tablet with enteric coating might get through the stomach acid with less loss of TTFD. He said that he had a tablet with a small dose of lipoic acid in it and we both agreed that since it was a colleague of thiamine it would do no harm to add the TTFD to it. This was before it was known that lipoic acid could be detrimental to the therapeutic benefits of TTFD and the dose is so small that I doubt it has very much influence.
Oh, ok got it. I thought it was a ratio based on science. Thank you for the clarification.
Michael
Great to know I was taking 600 mg of ALA. So the recommended ratio would be 7.5 mg for every 50 mg of Allithiamine?
I’m taking between 200-300 mg a day of Allithimaine. Having settled on the exact dose yet.
Best Regards,
Michael
Dr. Chandler Mars:
that is interesting as I have been taking much higher dose ALA also.
“…all sorts of problems including leaching thiamine.”
in brief what sort of problems?
thank you
Bernard Stricker
Besides the thiamine issue in someone who is already low, it can induce oxalosis – the build up of oxalates. This is partly related to the lack of thiamine and some other nutrients, but ALA can instigate as well.
Dr. Mars & Dr. Lonsdale:
I appreciate your explanation on ALA.
I purchased your book and am working on getting through and understanding the subject matter.
It is really incredibly helpful and appreciate both your considerable work! It is best the best explanation (cause and symptoms) and treatment of what I experience!
You are doing a great service to humanity!
Thank you
Thank you
Thank you. Please leave a review on Amazon when you finish reading it.
Hi Dr. Marrs,
Thanks for your reply! When you say an equivalent dose of ALA, do you mean equivalent to the TTFD or to the dose they used in Lipothiamine 7.5 mg for every 50 mg? I’m assuming the latter but just wanted to be certain.
I did start already on Aliithiamine as soon as I ran out of Lipothiamine, just wasn’t sure it would work until your clarification.
Thanks.
Michael
I meant equivalent to what you were taking – or less. It becomes a very high dose of ALA if one is taking a high dose of lipothiamine. There are mixed reports on what is an appropriate dose. I prefer a much lower dose at ~100mg.
So there’s no exact or approximate ratio to use as a rule of thumb? (Lipothiamine used 7.5mg ALA for every 50 mg of TTFD ) . So in the case of supplementation with between 100-250 mg of Allithiamine you’d recommend a general dose of ALA of about 100 mg?
Thanks again.
Michael
Lipothiamine and Allithiamine are different forms of TTFD and have the same action. The enteric coating is a refinement but not to a necesssity
Got it. Thanks for the clarification!. Regarding ALA my current understanding based on Dr. Marrs’s replies is that for a relatively high dose of Allithiamine such as 200mg daily or more, 100 mg of ALA is roughly the correct amount if one is going to take ALA , and probably should not be exceeded.
Would that be correct?
Thank you !
Michael
My 42 y/o daughter has alcohol induced thiamine deficiency. They gave her some over the counter thiamine and it is not working. I am now giving her sublingual. Would the TTFD be our best bet? She also has fatty liver and neuropathy. Where can I purchase this? Would like to see reversal of the symptoms.
TTFD would indeed be the best bet. There has been a big run on TTFD, I believe that Allithiamine is available from Ecological Formulas in California. Read about ” refeeding syndrome” on this website.
Hi!
I’ve had POTS (and most recently CFS) for 6 months now. Prior to these 6 months, I’ve had a horrible history with processed foods (never cooked at home unless it rhymed with Pizza Rolls or something microwavable), sugar (used to drink a gallon of orange juice a day, with cookies, and more recently (this year) caffeine (had an energy drink phase for a few months, 200-600mg of caffeine per day).
My doctors all think I’m fine because my whole blood thiamine is actually on the higher end of normal and pretty much every other blood test is normal.
Symptoms:
1. POTS (not typical POTS, don’t get dizzy, just get uncomfortable and very high heart rate when standing or moving positions)
2. Chronic fatigue syndrome (I’ve been experiencing “crashes” after light exercise, any idea why this happens, cellularly?)
3. Swollen/enlarged turbinates
4. Severe muscle weakness/fatigue (mostly arms and legs)
5. Sleep problems (I wake up constantly throughout the night)
6. Peripheral neuropathy (right hand and foot get pretty cold)
7. “Burning” sensation (lactic acid buildup I believe)
I came across your website at my last ER visit and they told me it was impossible that I could have beriberi. I took 500mg of Benfotiamine that night and my heart rate was much, much lower, I felt very tired but noticeably more calm, and the effect wore off just before bed (lucky me). I took 750mg the next day and noticed a heavy tiredness again, but the heart rate dropped once more, and I got great sleep that night. The next few days were similar – lower heart rate, less interrupted sleep. I started slowly taking less Benfotiamine and more Allithiamine (waiting for Thiamax in the mail) and noticed my symptoms coming back again and my sleep worsening. I experienced a crash soon after this when I went to physical therapy (symptoms all worse again the next few days). Since then, been taking 600mg/day of Allithiamine and am noticing a few hours after taking it my heart calming down and my heart rate being much lower than normal when standing. I take a b-complex 50 now, along with 400mg of magnesium and 200 coq10.
Whole blood levels of thiamine 5 days after being on Benfotiamine were in the 800s. Before taking Benfo, in the 150s.
Questions:
1. Is it safe to be taking as much as I am, given the blood levels? I know they aren’t well correlated, but I don’t know what to say to my doctor at this point other than “it seems to be working?”
2. Why might I keep experiencing these “crashes” after exertion if my body is supplied with enough thaimine?
3. Is what I’ve mentioned here indicative that thiamine is my issue? I’ve never noticed my heart be this calm, especially standing, but the results seem on and off. Sometimes it kicks in, sometimes it doesn’t, so I keep increasing the dose until I get this effect. My paradox reactions seemed to be worse the first couple days, and manageable afterwards.
Thanks so much!
Please understand that our use of thiamine in megadoses is very new and is nothing to do with vitamin replacement. Thiamine is being used as a non-toxic drug and there are still many questions that surround its use. You report that the blood level of thiamine is high and that that is the reason that the doctor has rejected the idea. Let me try to explain this. There must be an appropriate ratio of calories to thiamine. If you indulge in empty calories as you admit in your report, you distort the ratio. In other words the blood thiamine level would have been perfectly adequate for a healthy diet. This abnormal ratio has existed for a long time and correcting it requires patience. The first thing is to adopt a healthy diet and the second thing is to continue with thiamine supplement plus magnesium and exercise patience.
The blood level of thiamine is virtually always normal in mild to moderate deficiency. It only becomes abnormally low when the deficiency is severe. The only thing that you can do is to press on with the allithiamine and titrate it to the symptoms. Add B complex, magnesium and a multivitamin.
Cam,
In relation to your issues above – 2,4,5 7
Particularly the muscle burning in your legs, did this start to improve at 600mg? How are you doing now.
I’m on 350mg so far after 3 months. Going to aim for 600mg to see if this improve
Dear Dr. Lonsdale,
My MD suspects that I may have a case of Wernicke’s encephalopathy upon conveying to him some balance issues and memory issues which recently developed that subjectively seem significant. (the balance issues seem to come and go however) . The diagnoses isnt very obvious or clear cut but he suspects it strongly for a few reasons. He recommended I take Thiamine HCL and a Multi. . I saw my Neuro-ophthalmologist yesterday (I am seeing him for Papilledema that Tahnk G-d has gotten better, which he and other doctors attributed to Idiopathic Intracranial Hypertension). He too reccomended Thiamine HCl to start now and has reffered me for a mri.
I have heard that it’s used frequently for this condition, however all the literature I have been able to pull up seems to be saying unequivocally that initially the b1 must be delivered intravenously or intramuscularly I think because thiamine HCL isnt very bioavailble even at high doses.. He hasnt given me a good answer as to this contradiction and was wondering what you thought and what would be the most beneficial form of b1 and dosage for suspected early stage Wernicke’s encephalopathy in your opinion? Your input would be greatly appreciated.
Thanks!
Simon Nathan
You are very lucky to nave a doctor who suspects Wernicke. In 80 %of cases it is missed. The standard dose of thiamine I/V is 400 mg, repeated, because it is a disease long in the making. I presume that you are not an alcoholic or you would have mentioned it. The best oral thiamin is Lipothiamine, available from Cardiovascular Research. You will need 1-200 mg/day. Be sure to add 150-300 mg magnesium taurate and a healthy dose of B complex. Be aware that a blood test for thiamine could be normal
Dear Dr. Lonsdale,
Thank you for your reply and advice. This particular doctor is a top Nephrologist here in NY and he’s also been a close friend since I was in my teens so there were a few advantages talking to him on the subject. In terms of alcohol consumption, about a year ago, for the first time in my life, I start chronic drinking. I stopped for 3 months after the Papilledema diagnosis, but then about 3.5 weeks ago started again, this time consuming much less alcohol. Around 2 weeks ago is when I noticed the memory and balance issue. I have stopped drinking again.
Re Lipothiamine, would that still work at that dose and once daily considering the alcohol history? Additionally, I did read that there were some safety concerns or adverse effect possibilities for certain people taking it, especially if your GSH is low.
Would Allthiamin also work for this condition and if so at what doses and how many times daily?
Thanks so much for your help on this matter. It seems like you are the only MD I’ve encountered who understands the subtleties involved with Thiamine and other nutritional issues. Your work undoubtedly is helping many people.
Sincerely,
Simon
The history of alcohol makes all the difference. I recommend that your physician looks up the extensive literature on the treatment of Wernicke
The history of alcohol makes all the difference. I suggest that your physician looks up the extensive literature on treatment o Wernicke
Dear Dr. Lonsdale,
Neither physician is sure. In the case of my friend, he went from a strong suspicion to feeling it’s just a possibility after thinking it through. He didn’t examine me, it was a phone conversation. He now feels that it may just be b1 deficiency considering the alcohol use was roughly less than a year and I had stopped for 3 months before and was asymptomatic those 3 months , and just presented symptoms after 2 weeks of returning to moderate consumption.
I didnt present with confusion,delirium, and other symptoms he’d expect and there are other diagnoses that may fit better considering my I.I.H and the meds I am on. Nevertheless, I am going for the mri and seeing another neurologist to sort it all out.
I went through the literature connected both with Wernickes and alcohol and Wernickes not induced by alcohol with his brother (also an MD) but not as familiar with the condition and he was surprised that what he considered the “solid studies and guidelines” all concluded that the b1 had to be administered via i.v. or intramuscularly and yet many doctors treat it with oral Thiamine HCL, apparently even in his hospital which is a prestigious one.
That having been said I did find a few small studies showing that oral administration with allithiamine had the same effect as i.v/ i.m B1 in treating Wernickes. I think I also found one with oral TTFD/Lipothiamine eliciting the same results.
Since everyone including the MD’s are confused by the administration of oral b1 , I was wondering if you did consultations over the phone which I’d, of course, be happy to pay for, and if you didn’t if you could refer me to someone you trusted on this issue that did? I am at a loss on how to proceed with no proper guidance.
Thank you so much for your help on this matter.
All the best,
Simon
Let’s be clear. Wernicke is merely an extension of TD. Originally thought to occur exclusively in alcoholics it is now known to exist in non alcoholics. The whole picture is seldom seen, making it a difficult diagnosis. There is no criterion for the amount of alcohol that gives rise to Wernicke . Your MRI may help, but even if you might be wrong a clinical trial with megadose thiamine can not do harm and might be the final proof by recovery. It does not need high tech medicine. All it needs is clinical suspicion based on a pepared mind and a brief literature search.
Hi Dr. Lonsdale,
Understood and agreed.
Should I still take your recommended dose of 100-200mg Lipothiamine in addition to Magnesium Taurate or would the dose now be different?
I purchased the Lipothiamine brand you suggested (Cardiovascular Research) and also bought Allithiamine caps by Ecological Formulas. If I end up taking the latter for the long run, do I take the same dosage you recommended for Lipothiamine?
And do I continue until certain or all symptoms remit?
My balance is pretty decent, just a bit more unsteady once in a while but my memory seems to have deteriorated. Thnk Gd not anywhere near K.S. but enough to concern me.
If it is W.E. is it still possible to reverse or mitigate/halt the memory/cognitive deterioration using the protocol two weeks after the symptoms started? (which is about now)
I ask because the literature seems to say “no”. In particular, I found this review of studies that seemed comprehensive (to me, a layman at least) and although it’s mainly about K.S., there’s a section about Wernickes alone stating “W.E. can be treated effectively by the intravenous or intramuscular infusion of large quantities of thiamine.52 When this is done promptly, that is, within hours after the development of the encephalopathy, a full recovery is likely to occur. Even after a few days have elapsed, a complete recovery is still possible after thiamine replacement. When treatment is further delayed,……(the continuation is pretty disturbing – to paraphrase “, it’s not really reversible, K.S. will probably follow and irrespective of K.S. following or not it will leave residual damage to the brain etc etc ) 33,53.
Was wondering what your take on that prognosis is and is memory and cognition still recoverable after 2 weeks of mild-ish but concerning symptoms using the supplements, you suggest in proper dosages?
Study-
Korsakoff’s syndrome: a critical review :
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5708199/
Thanks!,
Simon Nathan
Hi Dr Lonsdale. I became toxic in b6 and also had a very bad reaction to acyclovir. Doctors are thinking fibromyalgia, CFS/ME. I have Extreme fatigue, neuropathy, brain fog, and muscle pain that is worse from exercise. Your article and everything in this site is fascinating and gives me hope. I recently tried magnesium malate and had a bad reaction– almost passing out. I just started thiamine mononitrate 100mg. So far so good. I have the lipo form on the way. Is there a time it might be safe to re-introduce magnesium? Thanks!
Hi thank you for being so gracious in sharing your knowledge. i have an ileostomy so taking oral supplements is of limited benefit. i now have neuropathy, extreme fatigue and muscle weakness. My doctors tell me I’m lucky to be alive and to lower my expectations . I have low b1 and b12, and clear nutrient deficiencies. I’ve begun iv 200mg HCL B1 daily for the past two weeks and noticing a benefit. I’m wondering if there are transdermal or sublingual products in order to ensure absorption? many thanks Sasha
You should try Allithiamine (powder) or Lipothiamine (enteric coated 50mg tablet). Both are thiamin tetrahydrofurfuryl disulfide (TTFD),so called open ring derivative of thiamin. It is more easily absorbed into cells than thiamin and you can titrate the dose to symptom improvement. It is non toxic.
Thank you Derrick, i cant take enteric coated tabs due to ileostomy but will certainly try to powder as suggested. many thanks for your help, Sasha
Yes, thank you for all of this information! I am wondering where to purchase allithiamine powder. I can only find it in capsules, but I’m looking to mix it in water for a feeding tube. In my experience, opening a capsule and trying to use it this way doesn’t work. Thank you!
A similar chemical, also a thiamine open-ring disulfide derivative, sulbutiamine, is available as a powder through several online venders (like Nootropics Depot, to name one). I don’t know what the equivalent dosage to TTFD would be, however.
Dr. Lonsdale has said on these pages that a person can use that form as well and that it should work similarly, but that he hasn’t personally.
Personally, I am taking 50 mg of TTFD and 100 mg of sulbutiamine daily (as well as 150 mg of benfotiamine and 500 mg of thiamine hydrochloride). However, that doesn’t mean that would be right for you.
A typically sized sulbutiamine capsule contains 200 mg, for whatever that is worth. Is that because it’s often used at far higher doses than TTFD, that it’s a cheaper ingredient so they can afford to put more in per capsule, or that TTFD is significantly stronger per mg than sulbutiamine, I can’t say. Only that the chemicals should have similar mechanisms of action.
Perhaps Dr. Lonsdale has an opinion about sulbutiamine as an option or what a roughly equivalent dose of it would be compared to TTFD? That would be really helpful to know, if anyone does!
Hello, has anyone used the Lipothiamine transdermal cream that is available. I wonder if it could help those sensitive to eating sulfur
Thank you for such great information! I got very excited after researching B1 as it sounds like EXACTLY what I need, but then I realized it is a sulfur containing vitamin and I have an extreme sulfur sensitivity. My main offenders are garlic, onion, eggs, citrus. I was able to eat all of those in fairly large doses until about 7 years ago after some extreme stress and other health issues. I keep trying to figure out how to heal my body, but it seems like everything I try makes me worse off. My body hates transdermal magnesium – but I feel like I need it! I have a hunch that the B1 is the exact same story. Is there any way for me to take this without having the sever sulfur issues? Thanks, Courtney
Courtney, molybdenum helps as a cofactor in activation of the sulfur processing pathway. Sometimes low molybdenun causes sulfur sensitivity. You may want to get your levels checked by hair analysis. Or you could try a low dose 100 to 200mcg molybdenum glycinate supplement. Someone I work with who eats a usual American diet experienced low molybdenun detected by hair analysis and had sulfur sensitivity.
Low b1 can also cause some people to have unusual negative reactions to magnesium, because some of the reactions activated by magnesium also futher use up available b1. Watch eonutrition (Elliot Overton) on u-tube if you want to hear a lot about b1 and processing sulfites. He refers to doctor Darrick Lonsdales book alot on b1 deficiency. Best wishes
I suspect thiamine deficiency, but felt toxic upon starting Allithiamine. Been using Benfotiamine since. I suspected it may be a sulfur thing. Do you think adding molybdenum to my stack would possible relieve me of this issue with Allithiamine?
I don’t know
Grace, Thank you so much for the info I am excited to check out the Elliot Overton video you mentioned! I have mo-zyme by Biotics Research in 50mcg tablets but I have never taken more than just one at a time. I have been looking into hair analysis, I guess I should just DO IT already! I feel like my health issues just keep compounding and the more I try to research and figure things out the more complicated it gets. Finding a doctor to understands anything has been impossible. My homocysteine is 24 which is really high, but I don’t tolerate B12 which is what I know I need to lower it. I have always eaten very clean, cook from scratch, and am very, very active outdoors. The stress of the last 10 years with my husband being a Leukemia survivor (in remission but not considrered cured) with major effects of extreme radiation treatments I have dedicated 100% of myself to try to help his health improve and the constant stress has cost me my health at this point. I will continue to research and learn though!
I found Molybdenum (from vegetable culture) Mo-Zyme Forte, by Biotics Research to be the secret to sulfur issues. Other molybdenum helped, but the Mo-Zyme Forte was the magic bullet. I chew one before breakfast and now I can eat eggs and onions. Take one with lunch as well, but do not take after 1 pm, as it will give you energy. Take for 10 days before adding B1.
I take Sulbutiamine (B1), because the Allithiamine was giving me some sulfur problems. I order the Sulbutiamine from Double Wood directly. The Benfotiamine does not bother me, like the Allithiamine, but I do not take the Benfotiamine, because of the blood/brain barrier issues. Benfotiamine heals nerve problems and works well with diabetes. B1 and molybdenum are a miracle cure.
What were your issues with Allithiamine? I get toxic feeling and diarrhea. I do have a history of GI issues. I do not get this with Thiamine HCL or Benfotiamine. Was your Allithiamine experience similar? Do you think this is a sulfur pathway problem? I have molybdenum at home too. Considering trying Allithiamine again after 1-2 weeks of supp’ing the molybdenum.
Allithiamine can increase gut motility. Good for constipation, but maybe too good for those without.
I tried it and got really anxious and got cramps. I’m going back to benfotiamin which has no side effects for me
I have a big issue with anything high sulphur I have figured out. I cant tolerate Thiamax for this reason and not sure if adding Moly would help but I think Im going to try this because I miss onion and garlic lol
At Amazon, I can find allithiamine, Benfotiamine, and Lipothiamine but not Sulbutiamine. No Amazon vendor seems to offer it. But I do find Sulbutiamine at Google from vendors like SuperSmart and Noorotrpics with whom I am unfamiliar. Can you recomnend a brand name and the best place to purchase it from online? Thank you.
It can be ordered directly from Double Woods.
Hi…I don’t see any of those supplements at Double Woods. Am I missing something? Thank you!
To clarify, I meant the allithiamine and lipothiamine. I am looking for a powder to mix with water to put through a feeding tube. Thanks!
Allthiamine is in powderm form so you can easily use a feeding tnbe
Hello Dr. Lonsdale and Dr. Marrs,
Thank you for the work you are doing. I’m sorry if this is a redundant question but when you recommend taking thiamine and magnesium do they have to be taken at the same time or can the magnesium be taken at night and the thiamine at breakfast and/or lunch? Thank you so much!
They can be taken together or separately
Hi Dr. Lonsdale,
Firstly, thank you for the work you’ve done on informing the world about Thiamine and its derivatives. Your work is outstanding and has clearly helped countless people. Not only that, your work is clearly pushing a wave forward of new “medicine” that comes from nature and is focused on true healing, not fixing symptoms.
I’d like to ask you for a comment on my case so far. I’ll try to be brief but detailed, as I know you must be busy.
As a kid I was very active, tanned easily, had a lot of energy and pretty normal as far as kids go. In my country, prescribing antibiotics is normal enough for general problems (even when it has no effect), not sure it’s related but I thought worth mentioning as it’d have been prescribed to me a few times when growing up. Around the age of 12, I started to have issues with my gut. I would get intense abdominal pain and stopped going to school as a result. Shortly after this, I became very pale and very weak physically, I was far weaker than anyone else in the school by a significant margin and so pale that it everyone noticed and used to insult me over it. The changes were clearly very extreme and abnormal. Over the years after this, I gained weight and continued to be relatively physically weak and very pale (unable to tan). It stayed like this relatively unchanged until I hit my 20s. Worth noting, my diet as a kid was quite bad, it consisted of a lot of junk food and soda, as I was very fussy with food. It was difficult to get me to eat meat/vegetables and I never consumed fruit. This was less a case during my teenage years and not at all into my 20s. Also worth noting, once the stomach issues began, I became heavily constipated and had consistent issues with digestion ever since, particularly it seems with stuff that are very “doughy”, like pizza would have a high chance of triggering abdominal pain. At some point in this timeline, I was also prescribed PPIs which I used for over a month for acid reflux issues (PPIs are horrendous of course, I know that now).
In my early 20s, I decided to try and get fit. I lost a lot of weight by dieting and exercising. I started lifting a lot of weights and using various preworkouts and supplements (not thiamine) to gain energy for workouts. The preworkouts easily contained 300mg of caffeine. I always felt like I struggled with the workouts, however, then I starting having an issue which I see other people refer to as “air hunger”. This issue would come after a heavy workout and persist for a few hours. I just thought I was tired, so I continued working out, with less intensity and more breaks. However, there became a point where the air hunger didn’t go away. In fact, I would be struggling to breathe and feel satisfied by breathing for days. I would lay in bed at night simply gasping for air, trying all manner of tricks to get “satisfied”. It was as if I could not expand my lungs to take in the level of air I wanted. I went to the doctor and he claimed it was a “muscle injury” and to wait a while. I did wait a while – no improvement. So I went back, the doctor said I was having exercise induced asthma, without testing me and prescribed me an inhaler. Using this inhaler made no difference, so I immediately stopped using it and went back to the doctor. They then did a lung function test, which came back perfect, in fact, quite a good score in terms of lung function. The doctor then essentially (without saying as much) said it was probably psychosomatic and stress related. I like to think I handle stress quite well and have done my whole life, very little affects me really.
Frustrated with all of this, I spent 2 years studying varying aspects of nutrition, vitamins, minerals etc, merely searching for answers. I tried various diets without much results. Sometimes there would be improvements, but often not. Over the years, the symptoms lessened but persisted to an extent occasionally. I did numerous blood tests, urine tests etc, all of which came back relatively normal. One odd note was that pyruvate and lactic acid on a urine test came back as low, which is the opposite of what you’d expect with a thiamine deficiency, right? On the same test, the lab (Genova Diagnostics), suggested my data required a higher need of specifically Zinc, Magnesium and B6. I found this rather odd, as these were supplements I was taking during my hardcore fitness fiasco. How could I require those vitamins, and why those in particular of all vitamins, the ones I was taking, would be highlighted in a urine test years later?
Then I discovered the YouTube videos of Elliot on YouTube, where he goes through a lot of your work along with Dr. Mars. Interested and with a why not attitude, I acquired some Alithiamine. I took just one capsule of Alithiamine at 50mg for a couple of days and immediately I noticed one very clear change, my digestion had improved enormously. First, I got very loose stools, which is better than being chronically constipated if you ask me. I also noticed that actually I had some “POTS” as I hear it described here, maybe a little bit of weakness etc. However, I knew about the “paradoxical” reaction as you call it, so I kept on trucking.
Continuing to take the Alithiamine, after a few weeks, my stools just became what I would call “perfect” bowel movements, consistently. I was astonished. I also tended to feel a bit more energy and have not really felt any “air hunger” to an extreme level in quite a while. I continued taking it for a few months at the same 1 capsule 50mg dose until then I stopped taking Alithiamine for a little while to see what would happen and I noticed a slight increase in “air hunger” and constipation after a week or so. So I started taking it again and I noticed the symptoms decreased again. I am looking for the possibility of being able to do the ETK test to confirm a diagnosis but it is difficult here, so it may not be possible.
So my question is, does this sound like thiamine deficiency that started a long time ago to you? What would be your recommendations in this case in terms of taking the Alithiamine. Should I increase dose? Should I try to lower the dose? Should I be able to reach a point of not having to rely on the supplement in the future?
Thanks for any feedback you can provide and for your work thus far, it is truly astonishing what you have discovered.
Yes, it does sound like longstanding thiamine insufficiency, possibly other nutrients as well. If 50mg of Allithiamine is working for you, keep at it. Some folks find that with additional life stressors, common illnesses, etc. that they have to up their thiamine for a period of time to meet the additional energy requirements but then can lower back to their maintenance value once it is resolved. Given the long term nature of your health issues, you will likely need thiamine indefinitely.
Same here! “physically weak and very pale (unable to tan)” I have wondered about that – this happened to me too, from puberty on aprox age 12 I no longer got a tan..
Also never gained muscle properly, despite of being quite into sports.
2007 burnout, Histamine intolerance (possible MCAS) and also exercise intolerance..muscles feel kind of sore and heavy and painful after very little exercise, walking upstairs will feel very exhausting in calves and I have to stop and let muscles recover… more than 15 min exercise will result in fatigue/exhaustion, red head/flushing/hot flashes and migraines…
As a kid until 2007 high sugar consumption…
Vegetarianism over 19 years
Veganism 4 years
My body does not react to well to Benfothiamine, but I take high doses of Thiamine Hcl
For some reason my blood work now shows low molybdenum and selenium, even though I eat eggs daily and am practically zero carb. Indication to leaky gut and they said something about kidney function not right..
What are your thoughts Dr. Lonsdale and Dr. Marrs?
Ps: I could not find much on kidney function in your book…but from Dr. Jason Fung and Dr. Berry I know that high protein and high animal fat consumption does not harm kidneys, it actually improves function..
No harm in trying. Thiamine is non toxic but you have to be aware of “paradox, also known as refeeding syndrome
Astonishing that you got such lousy medical “answers” Your dietary delinquency in childhood set you up for life. I totally agree with Dr Marrs and I find this commonly repeating case report very saddening. We are supposed to have solved medical problems with more and more technology and seem to have “missed the boat completely” by ignoring the biochemistry that makes us tick.
Thanks for your reply Dr. Mars and Dr. Lonsdale.
Another question, do you think it is possible to repair the defective enzyme? You say that if we have these severe long term symptoms and long term chronic deficiency that we’ll likely have to take it indefinitely, however, why is that? Is there not a way to fix it properly?
I find the idea of having to rely on supplemental vitamin to stay healthy is also not ideal. I would far prefer if I could fix the problem. It seems that, just taking the specialized supplement in this case doesn’t solve the issue permanently, even when the levels in the body should in theory be restored to full capacity. Why is this? Is it a defective enzyme due to life long damage?
Is there a way to fix this?
Forgive me if I sound “pushy”, I’m just trying to understand the underlying cause of the nutritional requirement for such high amounts of a vitamin permanently. It’s something I’d like to see a real cure for, which lasts.
Dear Dr Lonsdale,
I am a 55 year old female and have suffered from chronic migraines since my first IVF treatment in which I was given a spray version of Lupron (done in the U.K in 2001). I did another round of IVF in 2007 (injections daily of Lupron for a month this time) and since then, my life has never been the same. I am constantly thirsty and urinate frequently, which does affect my sleep a lot. I am always tired. I never did manage to conceive and have gained at least a 14 pounds in the last few years. I have not been able to work since 2007 as I’m in daily pain. I have seen an endocrinologist for at least the last 5 years as well as numerous other doctors and none of them have been able to help. I have the MTHFR gene permutation CT677. I can’t take B2 or B6 as they both keep me awake. One of your other correspondents mentioned SIDS – my father’s brother died as a baby from what we assume was SIDS. I wonder if Lupron has caused dysautonomia. Do you think Lipothiamine could help? I did contact Dr Marrs and am very familiar with her Lupron pages. Thank you for your assistance.
Kind Regards,
Belinda Fagan
Hi Belinda, Dr. Lonsdale is unavailable at the moment, so let me answer. Yes, thiamine would help. Thiamine is critical for mitochondrial function and Lupron damages the mitochondria. Recovery depends upon supporting mitochondrial recovery. Lipothiamine is a good choice and should help you to tolerate the other B vitamins better. Without thiamine, the other B’s are not utilized properly. Thiamine requires magnesium, so include a magnesium supplement at 100 -200 mg. Personally, I use a magnesium glycinate formulation. Others prefer magnesium taurate, threonate or malate. Regarding the lipothiamine, start at the 50mg dose, and titrate up slowly over the course of weeks. Read up on what Dr. Lonsdale calls a paradoxical reaction and refeeding syndrome. Sometimes long term or serious deficiencies cause symptoms to get worse before getting better when thiamine is initiated. As you investigate these, be sure to read the comments sections in the various articles that discuss thiamine. To navigate those, just reduce the dose and titrate up even slower. They resolve over time. How long this takes is entirely individual.
Hi Dr Marrs,
Many thanks for your speedy reply – it’s very much appreciated! We have corresponded before regarding Lupron and the truly appalling effects it has on the body.
May I make a suggestion? You and Dr Lonsdale are the only people I have come across on the internet who discuss Lupron and its horrific effects, apart from Lynne Millican. Without you people, I would have thought myself crazy. I do so appreciate your help. Perhaps you could invite other Lupron survivors to talk about what helped them and maybe there could be a discussion about thiamine supplementation, its co-factors and if it helps. Previously, I have not been able to discover a way out, after 13 years of living with daily migraines, thyroid and cognitive problems and other awful symptoms. I have been supplementing with iodine and selenium for the last few months. They seem to helps bit but now I know why I still feel so cold. (It is winter here).
I notice one of the other ladies on the blog suffered from chronic migraines and yesterday ordered the supplements Dr Lonsdale suggested – the Lipothiamine and magnesium taurate. I will report back once I have been going for a few weeks.
This advice about thiamine supplementation has given me hope that perhaps I may find some level of improvement to my failing health. I was always a fit, slim and healthy person before Lupron and its effects have been utterly devastating, mentally and physically. I literally feel that nobody, even my family and friends believe what I have gone through. It has been a very lonely experience.
I really do thank you both for the sterling work that you and Dr Lonsdale are doing- you are marvellous!
Many thanks and Kind Regards, Belinda Fagan
I am glad it has given you hope. It should help quite a bit. It may not be immediate, as these things take time, but it will help. As far as inviting Lupron survivors to post – I would love that. Periodically, I will post on the various Lupron pages, but I feel like I am being intrusive, so I don’t do it that often. If you know of women who might be interested sharing their stories, please have them reach out to me. Health stories are critically important to building a knowledge base around this and other drugs.
I am wondering if there is a connection between low thiamine levels and low GFR. Most of the women in my family have low GFR. My sister was told it is Berger’s disease ( IgA nephropathy) but nothing to worry about at the moment since GFR levels may be low, but are stable year to year. I started taking Lipothiamine in January for some other issues. My levels were at the bottom of normal. Just got lab results that showed my GFR increased from 51 a year ago to 63 now. Wondering if there is a connection and, if so, whether the need for activated thiamine could also be hereditary. Thanks so much
No question that there is a connection, so keep up the thiamine and don’t hesitate to increase the dose.
Dr. Lonsdale,
A couple of quick questions from some autism moms. Is it ok to take allithiamine with food, or does it need to be taken on an empty stomach?
We are beginning with 50 mg and stepping up to 100 mg, which from your books, seems to be the recommended long-term dosage. What would be some indications that more was needed by any given child, or should the 100 mg of allithiamine eventually resolve the deficiency? Should we just sort of play with the dosage and see?
A few of us were familiar with your work from perhaps 10 years ago, and several experimented with Benfotiamine at that time. While some saw some good gains from that, I feel that my son (now in his 20s) is seeing spectacular gains from the allithiamine after only a week or two–a much more obvious response than we saw with regular B vitamins or the Benfotiamine.
Because the allithiamine seems to be more effective, we are wondering if there is a problem with cutting the tablets in half or quarters, starting with a very low dose in order to avoid paradox (especially for younger children). We thought this might yield better results than using another form of thiamine. As we understand it, the tablets are just an enteric-coated version of the Ecological Formulas allithiamine, and while we’d be losing the benefit of the coating, we would be retaining the benefits of allithiamine.
I am really enjoying your new book–though as a lay person, it is taking me awhile to work through it. I feel thiamine might be THE most overlooked issue for autism spectrum kids. Thank you!
Hi Amanda,
The lipothiamine tablets can be cut and divided if necessary. The allithiamine comes in a capsule and cannot be cut and while the powder can be emptied and taken in smaller doses, I understand that it tastes horrible and is just not consumable on its own or in a food. As far as the dosage, that varies highly by individual. Some do quite well on 100-150, while others need significantly more. It depends upon a number of factors, genetics, epigenetics, other conditions, severity and chronicity of the deficiency, to name just a few. There is no way to tell how much any one person will need and for how long. We typically recommend beginning at a lower dose and titrating up gradually over an extended period. Note the symptoms along the way. What is better, what is worse. Note also that additional nutrients are often needed, magnesium especially. If you have our book, take a look at the graphic in chapter 3. It shows what nutrients are needed for mitochondrial functioning. Not all will require supplementing, especially with a good diet, but it is possible that others will be needed.
Thank you! We will just experiment then. I do have the book and will study the graphic in chapter 3. Yes—that was the problem; the allithiamine powder in the capsules tastes TERRIBLE! Bad enough that the moms in our group were not able to hide the taste in food. The tablets are a real help, as they are small enough for young children to swallow along with applesauce or whatever. Much easier than the capsules.
Is it ok to take the allithiamine with food, or does it need to be taken on an empty stomach?
We are seeing great things in my own home with five people trialing the allithiamine, after only two weeks. Better every day! Thank you, thank you for your new book!
Yes, it is fine to take allithiamine with food.
I am glad it is helping. It such a simple solution but so often missed.
Can you please tell me the name of the book/books you are referring to please. Thank you.
Thiamine Deficiency Disease, Dysautonomia and High Calorie Malnutrition.
Hoping to get a little clarification on the difference between Fursultiamine, Fursultiamine HCL & Allithiamine from a supplementation standpoint.
I have been taking Allithiamine from Ecological Formulas and it has a very profound taste (even in the capsule) and I’ve noticed some issues with body odor that I have never had before.
I was curious if moving to a Fursultiamine product would offer some improvement in those specific areas?
Could you clarify which thiamine product from Ecological Formulas you Recommend? There are two.
I have hypothyroidism and asthma as well as my 13 year old son.
Thank you.
Lipothiamine is probably the best. Your asthma may respond as well as hypothyroidism. Both are from energy deficiency, but that is highly technical, so just accept my word.
Dear Dr. Lonsdale,
I have recently started taking Thiamine HCL after reading about its benefits for chronic fatigue. I also have irritable bowel syndrome, and this form of thiamine seems to be prompting diarrhea. I’d like to try Lipothiamine but I have read that it’s derived from garlic, which normally gives me diarrhea. Do you know if the Lipothiamine has enough remaining garlic to cause problems for someone who reacts to it?
Thanks for your extensive postings on thiamine.
Lipothiamine is NOT derived from garlic. It is a synthetic molecule of a thiamine derivative. Tolerate diarrhea if it occurs because that is probably an example of paradox that you can read about on Hormones Matter
So Lipothiamine is different from Allithiamine?
Can you clarify this for me?
Would lipothiamine be better for someone with possible sulfur issue? Or is it equal either way since both are TTFD?
Every time I take TTFD, my GI system is bad. Diarrhea, toxic-y, sulfury. I do not get this with HCL or Benfo.
Thanks!
Both are TTFD. Lipo is an enteric coated tablet. Alli is in powder form and has a vile taste.
Thank you so much for the work that you’re doing. My husband is 41 years old, and has been suffering with what seems to be CVS (Cyclic Vomiting Syndrome) for the last decade. Maybe longer, but that’s when we noticed that “it’s not another stomach bug.” Thiamine deficiency for him makes perfect sense. He grew up eating copious amounts of sugar (a protruding mountain of added sugar coming out of the milk in his cereal, for example), and always struggled with his weight. He reached 300 lbs (he’s 6’2″) in 2003, but started “portion control” eating, lots of protein, and ate Slim Fast bars, plus worked out 2 hours a day; lost 120 lbs in 6 months. Of course, this is totally unsustainable, and once he stopped, he gained it all back. Not to mention he was still chronically ill. After we were married in 2004, we were very poor and survived off food bank items; a lot of granola bars, ramen noodles, canned rice, etc. I didn’t even begin whole food-type cooking until around 2011 when his health really began to deteriorate. A lot of doctors didn’t seem interested in helping him, or were overly concerned with his vomiting issue since he was so overweight. Finally diagnosed with Celiac Disease in 2014 via biopsy (after 3 negative blood tests over the span of a decade). He was also on PPI’s (proton pump inhibitors) since around 2007 for his chronic GERD, but was very concerned about long term use, and so stopped in 2014 and went back to using Zantac and Gaviscon as needed (right now Gaviscon; the Canadian version with sodium alginate). He started intermittent fasting in 2017, but was unable to stick with a ketogenic diet along with it due to strong carbohydrate cravings. Dropped from about 330 lbs to 300, but quickly gained it back as soon as he stopped IF. Then he began carnivore in April 2018 and was able to stick to it, and lost 80 lbs, A1C dropped from 6.4 to 5.1, and liver enzymes from fatty liver dropped and went back to normal. He is still carnivore, and it has by far been the most helpful diet compared to everything else (he previously tried GAPS, SCD, low FODMAP, etc). But he still has GERD, still randomly vomits and gets strange vertigo (with left-beating nystamus), random abdominal pain, alternating diarrhea and constipation, and random, asymmetrical swollen tendons and sometimes what seems like gout, and migraine and tension headaches (all stuff he had for many years); also a weird “aura” where he feels like he’s “behind himself” when walking. In September 2019, he had a two week cyclic vomiting episode (never had one that long before), and ended up starting the “mito cocktail” as advised by Dr. Richard Boles (coq10, B-100, l-carnitine). After a month of that and no improvement, he began amitriptyline. Now he’s at 75 mg. He seems to be vomiting less often, but the vertigo is still crazy at times, he still gets bad headaches, and he still misses a lot of work. And he gained about 30 lbs (which he doesn’t need! He needs to lose more weight!).
I happened to come across your work from someone on Facebook, and I bought Allithiamine a few weeks ago. It finally came in yesterday, and he took 100 mg last night with 300 mg of magnesium citrate (he’s been taking the mag cit for several months now). I read about the “paradox,” and if it happens to him, I hope he gets through it quickly. He’s such a mess, health-wise. He is so complicated, and after two gastroenterologists, a rheumatologist, two allergists, a dermatologist (he also has skin issues), and now waiting for a referral for a neurologist… NOBODY has been able to figure him out! And it’s Canada where it takes two years to see any kind of specialist; especially for him where they put him low on the triage because he’s not “wasting away.”
Hi Sarah, he can NEVER get better until he STOPS sugar and I mean it. Cyclic vomiting is a brain energy deficiency disease. Allithiamine will not work if sugar continues and you certainly should expect paradox. Tell us what happens. You are wasting time going to physicians. Thiamine deficiency” does not exist” for them.
I was giving his history on how he *used to* eat a lot of sugar. Not that he does not.
Stops sugar 100%? Or 99%? Because he is carnivore, which means he no longer eats sugar. At all. Not even sweetener. He eats 95% meat, and the 5% is cheese and eggs. With a little bit of spices, and maybe mustard and Frank’s Red Hot. He also drinks coffee with heavy whipping cream. So no sugar. But perhaps three times a year or so he might have a gluten free sugar treat. Will the treat a few times a year interfere with him getting better? What about dry wine?
Is 100 mg a day of allithiamine for him sufficient?
I have no means of telling the dose.
Coffee blocks thiamine absorption
I don ‘t know where you got that info. We know that coffee CONSUMES energy while thiamine stimulates energy PRODUCTION. There is therefore a theoretical possibility that one cancels out the other——but that is not a block.
I’ve read that coffee (and tea), among other foods, contains thiaminase, which inhibits thiamine.
Here’s a link to a Pubmed article on Beri-beri that mentions it. Would be curious to hear your reply Dr. Lonsdale.
https://pubmed.ncbi.nlm.nih.gov/30725889/
He has a migraine brain and therefore a problem balancing his electrolytes. Get onto the Stanton Migraine Protocol Facebook page and you’ll get help there.
can you take the time to look at Garrett Smith and nutritiondetective.com?
Garrett sent me here to better understand B1 while I am detoxing from Vitamin A toxicity and copper toxicity.
It’s all connected.
Hello Dr. Lonsdale,
I among many are trying to help themselves heal and feel better. I won’t go into my long health history in to much detail. I have Hashimoto’s hypothyroidism, I have worked on changing my diet a great deal to include more whole foods, I cook as much from scratch as possible but do still also have some prepackaged things as well. I am trying but with life being busy and just trying to make it through each day, sometimes the shortcuts are necassary. I have tried many supplements which are touted to help a person, which most seem to backfire on me and make me worse. I have struggled with fatigue, depression, on and off edema (non pitting) and weight gain and just an overall slow body function for years. My two daughters also struggle, one with OCD It would be WAY to long of a post to explain it all. I have read a few different books and have done alot of research on things to help me feel better and try to get an “optimal” thyroid dose. In growing up we did not have great nutrition. Always struggled for money and ate alot of meals based around carbs, cereal etc. and I pretty much continued that way of eating for my family after I had children of my own. Not really knowing any better I guess and assuming that the fortified food was just as good as the fresh. And it’s not that we had no fresh food, just probably not to the extent we should have. After reading into your research more I am coming more convinced that maybe alot of my symptoms and my daughters as well, are stemmed from mitochondria dysfunction and possibly a chronic thiamine deficiency which has now manifested in a state of malnutrition basically. One test that always shows up low in my liver testing is Alkaline Phosphatase in looking back over my medical records it has ALWAYS been below normal range. Dr’s I have asked about this have told me they don’t worry about the Alk. Pho test unless it is elevated. In the reading I have done, that low test result can mean hypothyroidism, low zinc, or malnutrition and I suppose other things as well. I have been treating the hypo, I have tested my zinc it was low normal (tried supplementing with failure, it makes me more tired) I am trying to eat better but this test just never improves. I am curious if this could have any bearing with thiamine deficiency. I have tried to use 150 mg benfothiamine in the past. It seems to make me feel good but sometimes a little off and kind of what I describe as “hyper” feeling and then I crash later in the day when it wears off. I haven’t continued with it on a daily basis as I am just a bit scared. I have struggled so much in the past couple of years with thyroid medication changes and hyper to hypo feelings, digestive issues etc. And my experience with supplements that may seem to help in the beginning ending up making me feel worse later down the road, I guess I am just gun shy to go full in.
If you are interested, here is a link to Isabella Wendtz’s blog where she talks about Thiamine and thyroid fatigue, and how she supplemented to help herself with it. https://thyroidpharmacist.com/articles/thiamine-and-thyroid-fatigue/
I am curious what your opinion is on what she says as far as the type of thiamine and the dosing regimen she suggests. She has also written a couple of books on Hashimotos which she has healed herself from (not just from thiamine but with many factors) I would like to give thiamine a good try for me and my daughters but want to go about it the correct way. Thank you so much for all of the information you have put out there about this. I hope to buy your book when I am able.
I will give you an analogy. A car has an engine whose function is to PRODUICE energy. This energy is passed through a transmission that CONSUMES it. Some engines have flywheels that prevent the engine from running away with itself. In the body, thiamine enables ENERGY PRODUCTION. Other vitamins are part of the TRANSMISSION and the thyroid, like a flywheel, governs the speed of the engine. Obviously nothing happens without the engine so thiamine deficiency can cause ANY symptom, simply because it is vital in energy production. Note that health is nothing more than the ability to produce and consume energy. The thyroid simply governs engine speed (metabolism). Take 100 mg of thiamine and 125-150 mg of magnesium taurate a day and read about refeeding syndrome, also called paradox, on this website. Paradox is temporary recurrence or exacerbation of symptoms that can last as long as a month or so.
Today is Sunday March 2. I have reread many of the sad stories told on this forum. I am increasingly appalled at the common health problems recorded here. You must ALL look at the high ingestion of empty calories that make up the American diet, particularly the so called “goodies”——-donuts, rich creamy cakes and deserts etc”. This induces a metabolic disaster where the calories overwhelm the ability of thiamine to burn them. Even if the physician thinks of a vitamin deficiency he might take a blood sample and send it to a lab that measures the concentration of thiamine. Of course, it is reported as normal!!———–conclusion “lady you are crazy. You do not have thiamine deficiency. This is America where nutrient deficiencies simply do not occur. You are neurotic. Go home and pull yourself together”. Naturally you trust the doc that you know so well. Your husband plays golf with him and you socialize. The fact is that you have read the statements on this forum and you are in possession of knowledge that your doctor totally rejects. You may have suffered so much and have been rejected by so many doctors that you are naturally suspicious of a potentially simple answer written here. All I can say is that many drugs precipitate thiamine deficiency and it can be the result of prolonged stress. I even think that minor genetic errors can magnify this deficiency. Call ICIM (International College of Integrative Medicine) or ACAM ( American College of Advancement in Medicine)for your nearest practitioner. They are still “rare birds” so you may have to travel.
Dr. Lonsdale,
Thank you so much for your reply. Without information that I have “googled” I think I would be a much sicker person, just following what most of the doctors I have seen advised to do, which is almost do nothing. So thank you for ALL of your information. Sure would be nice if all doctors would take a true, real interest in helping to improve the quality of life for people. Although I believe I have improved in some ways over the last few years, I just can’t give up for myself or my daughters. We want our lives back. It is SO HARD to get help when you have to stay within your “network” of clinics and doctors. I will check out the ICIM and the ACAM and see if there is anything we can swing in the future financially.
Thanks again, this is so appreciated.
Hi Dr. Lonsdale,
I forgot to add this question to my reply back to you. We have been using “Natural Calm” which is magnesium citrate. Not everyday but a couple of times a week. What is your opinion on the difference between the taurate you recommended compared to the citrate?
Both are salts of magnesium that enable the absorption of magnesium. the therapeutic component. Mg citrate is OK
Dr. Lonsdale–THANK YOU to you and Dr. Marrs, from all of us who have been searching for answers for months and years.
I believe I have Vitamin B1 issues, leading in 2013 to an involuntary 40-pound weight loss, very painful GI issues, constipation, coldness in my hands and feet (and chest), insomnia, tachycardia, and irritability. It’s as if my body thermometer has been turned down. I’ve been to lots of specialists and twice have been to a world-class U.S. medical institution for week-long tests and consultations, only to be given Miralax. I have ordered (1) your book and (2) both the Allithiamine and the Lipothiamine supplements made by Ecological Formulas/Cardiovascular Research (respectively)–and am looking forward to reading/trying them.
My question today is about our 7-YEAR-OLD SON. He was given Cefdinir in April 2018 for a sinus infection and has had daily, near-constant stomach pain ever since. He also began to exhibit nervousness and fears we previously never saw–asking questions like what if we (his parents) died, being afraid to be away from us in the house, etc. These are more than the usual child-becomes-aware-of-the-world fears and were sudden, coinciding with the Cefdinir. He’s also regularly cold, despite his skin feeling warm. In the past few weeks, he’s becoming more and more irritable. We’ve been to the usual cast of otherwise wonderful, well-meaning providers–pediatrician, pediatric gastroenterologist, etc. He was prescribed Miralax and VSL. We discontinued the Miralax on 12/23/19 (after about 16 months of use) and switched to magnesium citrate gummies, and after VSL recently was discontinued, have switched to Visbiome.
I’ve read recently that Cefdinir and other antibiotics deplete Vitamin B1 and other B vitamins.
I would appreciate your thoughts, including the following open issues/questions:
1. May I give our 7-year-old son either Allithiamine or Lipothiamine? If so, how much, how often, when (i.e., what time of day), and with or without food?
2. Because he’s young, I expect he won’t swallow capsules well. From your posts, I see that Lipothiamine is enteric coated and preferred but that Allithiamine can be divided if necessary?
3. How much magnesium taurate–and when/how often? [He’s been taking magnesium citrate gummies very well for about a week, so if mag citrate is an acceptable option, that would be helpful.]
4. Is the transdermal cream a viable option OR should we stick to either the Lipothiamine or the Allithiamine?
5. Any other supplements we should give him or consider?
Again, our sincerest thanks for your expertise and your continued work, even in retirement.
Dear Dr. Lonsdale, I have been researching the various B1 supplements because I have wernicke encephaly and was diagnosed a little over a year ago. I live in Mexico and they never gave me the Thiamine IM nor did they give me the right amount of oral thiamine (only 100 mg) initially. It was an acupuncturist who told me to go on 500 mg. Question, two months ago I started taking benfotiamine and I think it is helping a bit along with PT (I have trouble walking) but after reading about the TTFD, I am thinking it might be more helpful to be on that. Since I have to order this from the United States…what would the recommended brand I order along with the amount I should be taking daily? I still take a daily multi as well. This is a mysterious disease. The only person that I have been able to communicate with about this syndrome is a woman who is not alcoholic therefore leading me to believe that some people may have the disposition for malabsorbtion. I am willing to try anything…….a clinical trial other supplements, etc. I am basically very healthy and no one would even know anything is wrong with me sitting down but once I start walking, I look drunk without alcohol. Any advice, recommendations or positive feedback would be wonderful on this Christmas day. Thank you very much.
Dr. Lonsdale, My symptoms have been widespread over decades. I have recently read your book. What would cause a sulfur sensitivity from taking Allithiamine? I seem to be sensitive to sulfur and it builds up hydrogen sulfide in my stomach. I felt better on 1 dose of Allithiamine than I have taking regular thiamine mononitrate. I felt it in my brain and was more relaxed, but the allithiamine locked up my digestion. I wonder what is at the root of this sulfide problem? How to work around it? I will try Coenzymated B-1 as Cocarboxylase next to see if that will help like the Allithiamine did but without the side affects of the gut distress.
Dear Dr Lonsdale,
First of all, thank you for your amazing work, and contribution to these subjects, it truly is an eyeopener.
I never heard about paradox before, and I am happy to know about it.
Imo, it’s a bit tricky to really tell if some symptoms are paradox, and you should continue dosing or not, or you are doing something wrong, but hey, who am I to tell.
Anyways, I wanted to ask you something about Lipothiamine,which on some sites is listed to contain 143 mg of calcium, 10 mg of Thiamine, 10 mg, and 7 mg of Lipoic Acid, is this actually correct, or is it actually 50 mg of thiamine or TTFD?
If someone would have acces to Fursultiamine, and he would be wanting to take the same dose, could he than take 50mg of Fursulthiamine if he was taking 1 capsule of Lipothiamine, or should he take 10 mg of Fursulthiamine?
Can we assume Fursulthiamine is Fursultiamine hydrochloride or is there any difference?
What I really wonder is, what your current regimen is regarding supplements/vitamins, but it is up to you, if you wish to answer this or not.
Thanks in advance.
This question is a lot more complex than it appears. Paradox occurs when a person begins to take thiamine in large doses after they have been deficient, sometimes for years. Thiamine itself has no toxicity until the dose reaches astronomical levels. The reason for paradox is that the patient has been in a state of catabolic metabolism and is attempting to switch to anabolic metabolism too quickly. I want to emphasize that Lipothiamine is a trade name exclusive to one vendor. It sounds as though pirating is going on and epitomizes the state of the culture that we live in. The chemical name is thiamine tetrahydrofurfuryl disulfide (TTFD). Fursultiamine is TTFD and is another trade name. I suggest that you read some of the posts on “Hormones Matter”.
Thank you for your fast response.
I guess that that is the case for me, going from catabolic to anabolic, since I finally started to gain a little bit weight.
The problem is however, the increased anxiety, the low blood pressure, and the faster heart rate is hard for me to stick trough when taking B1. I was taking 100mg of Benfotiamine with 25mg of thiamine HCL.
Even when I open the capsule, throw away the half, I still often feel horrible.
The reason for me wanting to try Lipotiamine, is because it should be able to bypass the stomach, and get absorbed much better then conventional b1, and bypass issues like thiaminase. Or I have understood it wrong, but I take your word for it, that it is the best b1 to take.
I really hope it will help with my 99+ problems that doctors just can’t seem to diagnose.
Palpitations, random bouts of fatigue, frequent urination(pale), tinnitus that randomly spikes worse than ever, horrible memory issues, anxiety and feeling of getting a seizure sometimes after a heavy meal, bad concentraion, anxiety, bad mood, , low motivatio, exercise intolerance, trouble keeping up my weight. I am about 180 cm, and keep losing weight till I reach about 59, and than it stalls.
Maybe my past chronic alcohol use, my IBS has a role in it, I have my ups and downs, but I just can’t handle the stress of my memory anymore, and I am only 33.
I have not tried Lipotiamine yet, since the package is still on it’s way, but sadly, soon, is going to off the shelf, at the place I am able to order.
It’s kinda hard to get here, but TTFD is a lot easier to order, which to be honest, I was planning on doing so after about a year of Lipotiamine(that’s how much I have in stock once it arrives).
That honestly is the reason why I asked if 50mg of Lipothiamine would equal 50mg of ttfd, since some sites state that 1 pill of Lipotiamine only has 10mg in it.
I have read a couple articles, including the comments, and I am fascinated by the subject, and your input.
Can’t wait to get your book, and read it.
Thank you for your time, and sorry for my long post.
I thought I was reading the description of my own case… I have exactly the same issues. Weight loss in particular is nerve wracking. Almost same age too. Very tired of my medical ordeals. Hope we will find a solution!
Regarding the paradox reactions, I too had increased anxiety, faster heart rate (pushing towards panic attacks), and generally feeling awful. I was trying only 25mg Thiamine Mononitrate. The adverse reaction is quite quick to be produced (15-30 minutes after taking the supplement) and quite long lasting. Having read that the nitrate could be an issue, I ordered Thiamine HCL, planning to go very very slow on it because I am now a bit afraid of the reaction I have. But I tell myself there must be something behind that exaggerated body response and it give me hope as well.
Chris – did you ever get past paradox?
Unfortunately not. Had a really hard time and decided to halt for a while.
How long lasting was the paradoxical effects of the mononitrate and HCL forms for you? Did you have same experience with TTFD?
Hi Doctor Lonsdale, could you give examples of the types of paradox reactions your patients faced when taking B1 after being deficient?
This is a belated reply to Stephen W, Ann Knight, Kami and Cakir.The symptoms described by Stephen W include weight loss, painful gastrointestinal symptoms, constipation, cold extremities, insomnia, tachycardia, and irritability. His son experienced symptoms after being treated with Cildinir. These include abdominal pain, nervousness, phobias, and he constantly complains of being cold. These symptoms are typical of those caused by energy deficiency in the brain. The commonest reason for this is a high ratio of calories to thiamine. The reason that Stephen received MiraLAX for treatment is presumably because the physician focused on a single symptom, constipation. All the symptoms are because the brain is failing to adapt the body to the environmental situation of the moment. A boy of seven years can easily take 50 mg of Lipothiamine and 125 mg of magnesium taurate a day. My theoretical explanation is that there is a genetic component coupled with poor overall diet. Sugar precipitates thiamine deficiency and many drugs that are commonly used can also precipitate this deficiency.
Good Afternoon Dr Lonsdale,
I want to begin by thanking you for the wealth of information you have provided about this topic. My daughter has been through a nightmare with her health in the past 5 years. I will spare a lengthy description but to summarize : Her problems began shortly after receiving the gardicil vaccine. She subsequently contracted mononucleosis and was hospitalized for a severe case. Having never really recovered from the fatigue of mono her episodes progressed and she began to experience syncope, tachycardia, and shortness of breath with minimal exertion. Many and frequent trips to the pediatrician led to a referral to Childrens Hospital of Philadelphia where she was diagnosed with post viral POTS. She was treated for several years with minimal improvement and was left discharged from their care with the hope of “out growing” the POTS. At 19 she struggles with other strange symptoms including what seems to be carbohydrate intolerance and swelling of the feet and lower extremities. Quite frankly her story to me reads like a book for Thiamine deficiency and its related issues. I am writing because I have poured over the comments on this and other articles in the forum. I believe I have a grasp on the supplementation of Thiamin, but am seeking a guide for dosing and progression of the use of it. Are you able to guide me to a resource for this? I am a physical therapist and can understand complicated medical language. I just want to do this the correct way. The higher dosages are what concern me. Any guidance you can offer me would be greatly appreciated and again, I am so thankful for this information. Sincerely, Jodi
I am sorry. I didn’t see this until today. You need Lipothiamine from Ecological Formulas in California. Start with 50 mg a day together with 125 mg magnesium taurate. Symptoms will become worse (paradox/refeeding syndrome) for an unpredictable time, as long as a month or so.Gradual improvement will follow and you can then titrate the doses to the symptoms. There is no toxicity. Paradox is a switch from catabolic to anabolic metabolism. POTS is dysautonomia and is typical of thiamine deficiency. Discontinue ALL forms of sugar and sweeteners.
I assume when you say discontinue all sweeteners, this includes stevia and local raw honey, correct?
I feel embarrassed because others who posted after me are receiving answers while mine is ignored. Did I not handle my comment correctly in some way? I surely did not mean to waste anyone’s time, but I just wanted an opinion.
It may have just simply slipped through the cracks. With the volume of comments coming through, some are missed by mistake. If you would repost your comment, I will make sure that Dr. Lonsdale sees it.
Joan,
He did reply, but it was posted to the wrong thread. Again, there has been quite a bit of volume. If you scroll down, you will see it. Here is what he said:
Derrick Lonsdale says:
November 15, 2019 at 3:49 pm Edit
This long and complex story is absolutely typical of what happens when energy metabolism is defective. We do not know whether this is genetic in origin but I strongly suspect that the pregnancy had much to do with it. In other words genes do not usually work on their own. Diet is of immense importance for we are conversant with the fact that alcohol and sugar precipitate thiamine deficiency. Let us look at the history. He began seizures at the age of six months, suggesting a B6 deficiency. Extreme fatigue, migraine, broken bones, sugar craving, dilated pupils, low blood pressure, POTS are all typical of an energy crisis precipitating dysautonomia. That was why he stopped growing for two years. You should look up the post on the Hormones Matter about eosinophilic esophagitis because it illustrates the diversity of symptoms that develop when brain energy is defective. The best thiamine supplement would be Lipothiamine and start with 50 mg a day. Read about paradox as written on this website and wait for the paradox to subside before you start increasing the dose. Add B complex and a multivitamin. I would be interested in the treatment that he has been given for dysautonomia because that is clearly a mark of energy deficiency in the brain.
I am thrilled to get your answer and thoughts – it means so much. When you say the pregnancy had something to do with it, could you explain what you mean ( also, genes do not usually work on their own.). I may be a bit tired tonight, but I am struggling to understand that. As a toddler our son was addicted to sugar and he comes from a long line of alcoholics – me included. My problem was resolved 4 yrs before his birth. One set of his grandparents both were alcoholic and died from alcohol related illnesses. One grandfather on the other side had alcohol issues. Many great uncles, an uncle, brother, and sister had alcohol issues. I don’t know if this is important, but we lost 2 infants before this son; a daughter to SIDS and a son to Triasomy 18.
As our son grew we worked to reduce the sugar in his diet, but were not sugar free. From 2012 to 2014 he was on this regimen of sugary drinks and foods, recommended by a scientist and the dysautonomia symptoms ramped up from that, I think.
The neurologist handling his dysautonomia treatment has recommended compression stockings to waist, Midodrine 5 mg 3x a day, 5 mg of Propranolol 4x a day, salt intake of at least 5g per day, 2 litres of fluids, some Gatorade included, and a graded exercise program using a recumbent bike, rowing machine, swimming, or floor exercise. We are not able to do all this. He can’t drink that much, too much salt is excitatory, he cannot take Gatorade ( sugar free) or fortified foods. She advised eating every 2 hrs – he eats every 2.5 hrs and has for about 4 yrs. He cannot do the exercises often or consistently yet, but is moving more & hurting less. Midodrine helps SO much. He still cannot sit with feet down for too long. His blood pressures still stays on the low side. She also wants him in speech.language therapy, but we haven’t gotten that done yet, as he has been sick for nearly 3 weeks with a sinus infection. Oh, and he did have constant ear infections as a baby, got tubes in his ears, and then started having sinus infections.
Thank you SO very much for responding. It means a lot to us.
Hi Joan, Would you copy and paste this comment to one of Dr. Lonsdale’s threads, perhaps scroll down to his original reply to you so that he will see this. He won’t see it here, attached to my name. Thanks.
Dr. Lonsdale,
I am very interested in your website. My health issues started 6 years ago: tachychardia, dysautonomia (body temperature, can’t stay in the upright position for a long time) as well as some digestive issues (a lot of food sensitivities), brain fog, chronic fatigue. I had my fair share of examination that were all normal. My doctors have not been very helpful and my illness has been viewed as mostly psychosomatic.
Your website resonated with me because my diet was very heavily based on white rice (I viewed it as “safe carb”). I would say that at least 60-70% of all my food intake have been white non enriched rice.
I gave thiamine a try (Thiamine Mononitrate) 50 mg but after a couple of days, I had some bad reaction to the product (tachycardia, worst dysautonomia upon standing). I was surprised because the dose is fairly minimal compared to those of other patients (some take north of 1g). I was wondering if this is the kind of paradox you have seen in the past? Do you think it is due to the mononitrate part of the product or thiamine itself? Should I try thiamine HCL instead?
And thank you for sharing your experience. I can’t understand why such a central nutrient, low risk/high reward therapy is not being considered at all by the medical community.
Chris
You have classic beriberi. Wnite rice has been the cause for thousands of years Stay with relatively low doses of thiamine until paradox gives way to improvement, Add125-250 m g magnesium taurate ahd titrate doses of both to symptom improvement. Read the posts on this website
Doctor Lonsdale,
Thanks for your answer. Would you consider writing some time an article about the different example of paradox symptoms you have encounter over the years? I believe it would be very beneficial for people trying the thiamine therapy. In my case I feel like getting the thiamine in my system has an excitability effect after only 10-30 min after ingestion, maybe the effect thiamine has on acetylcholine?
And most importantly, I am very thankful you take the time to interact with me (us) though I am (we are) not your patient.
Thank you for writing this post. I have a long and complicated health history but I was hoping that diamine could relieve some of my symptoms. Obviously it won’t help all of them but some of the ones I was hoping it could relieve are my head pressure my dizziness my Blurry vision and maybe even my mild autism. I did have a few questions though. I started taking a alihiamine 3 days ago with magnesium I’ve been taking 150 mg pill and it has not helped at all. I’m wondering if I should have felt something? Another question I had was do I need to take it with a fat? I was also kind of concerned since I bought the supplement on Amazon I’m worried it’s not an authentic product. The label does not show the FDA disclaimer on it. There were only 245 capsules in the 250 bottle. It also does not have a garlic taste it’s more like a sulfur taste. I’m not sure how I would know if it’s authentic
Hello Dr. Lonsdale. In June 2017 I had septicemia, unkown origin, causing endocarditis and strokes from emboli. Eight weeks of IV antibiotics etc it was very difficult. Now I struggle with post stroke fatigue and live a very limited life, working some mornings but no travel, lecturing, public speaking as before. Symptoms: physical, cognitive &fatigue, easily sttessed, swollen tongue, dizziness. Am a 55 year old male on no medication. Might your thiamin, magnesium & multivitamin protocol assist? Thank you. Stuart
Why not try it——————————-BUT please read the information on this website about paradox.
Thank you Dr. I have since read your article on ‘paradox’ and saw the article on sepsis; very relevant to me (my infection was ‘staph aureus’)! I will pursue thiamine hci to start with, as I have some, plus magnesium & B complex or multi. As regards a multi, although I note various articles on Vit D on the website (in favour) do you have an opinion on recent discussion as to whether Vit D3 supplementation is (ultimately) immunosuppressive (due to being a secosteroid, whatever that is)? And even if it were, might that not be dose dependent? Thank you again very much, and all the very best.
Hello Dr. Lonsdale,
Our 28 yr old son’s story is very long – I’ll try to make it as brief as I can. Seizures started at 6 mos, just stares & muscle jerks, hypotonia alternately in sleep. These interrupted language development, but he can talk, just struggles ( not MR). Headaches at 5 yrs that caused him to sleep all morning. Partial blind spells at 7 with auditory aura plus spells that seemed like a complex partial ( I filmed it – 2 neuros said it was not a seizure spell), in that he couldn’t follow a conversation & would walk in front of cars etc… He was so tired during these spells & looked sick. 1st migraine at 9 & after that 3-7 per week for 8 yrs. Had 1st tonic clonic sz after starting Topamax at 12, stopped growing for 2 yrs, developed numerous nutritional deficiencies as time went on, 3 blood disorders. At 17 he spent 6 months being constantly ill with upper respiratory infections & was too weak to do anything but sleep. Migraines suddenly stopped & drop seizures and black outs began. Broken bones, injuries galore. Protective body & head gear became necessary. Severely underweight. At 22 had a hyperthyroid attack which set off deadly seizures – I began to work with an endocrine scientist who put him on a regimen of sugary milk, oj, ice cream. He loved it since that was all he ever wanted as a toddler on up. Thus began a really strange situation where sugar seemed to stop the seizures short term, but ramp up his instability overall. It took time, but eventually we realized sugar gave him hypoglycemia, starch without meat/protein produced reactive hypoglycemia. But he lost his ability to eat meat & many other things. His gastric emptying was either too fast or too slow & it impacted his blood sugar. He became mentally unbalanced, doing crazy things that nearly got him killed. But he had lost his ability to exercise and when he would try to run away from us he would fall and pass out & would need hours or maybe a day or 2 to recover. He hurt all over and felt broken. His eyes were dilated so they looked black. Eventually, we discovered he had low blood pressure all of the time and POTS. We tried a keto diet once & it seemed to nearly kill him by causing him to not be able to wake up for a long time of screaming and yelling, tickling, cool cloths etc… . He began to die, I think, in 2017 with chronic pneumonia. Every 3 months. The keto diet also took his ability to speak loudly enough to be heard away & it has never come back. He drooled constantly after the keto experiment. We finally found out about dysautonomia and are working with a neurologist via Skype. He has gained 32 lbs & is much better. We have a bag of tricks that help. But with a heavy heart I realized that just as I had always known there was something causing the seizures and I had to look for it, the discovery of dysautonomia was not the only answer. What was causing it? Why can he not tolerate sugar? He didn’t exactly fit the description of the few things that could cause such a rabid sugar intolerance. So, I stumbled onto this site and all I can say is, “wow”, it sounds like my Patrick. He had a b1 test recently, but from reading here I don’t think I should go by it. It was a LC/MS/MS type range 78- 185 and he was 127. I think if he has this it has been there since birth. Do you think it is possible he has thiamine deficiency? Which type of supplement would you recommend, if so?
I think I may have this, too. But when I take b vitamins I feel drunk!
Thank you kindly, if you read this.
I have answered this. Don’t know what happened to it
Hi Dr. Lonsdale,
I just wanted to give a quick update in case it’s helpful to others. I have been taking the cocarboxylase (co-enzymated B1) for a few weeks now and am definitely getting better! Energy and thinking is very improved. Muscle burn/pain is still an issue, but is a bit less, my postural muscles seem to be functioning better with less symptoms of my back etc being “out”. I’m able to be more physically active, but do still have abnormal shortness of breath periodically. Over all, there’s a great improvement and this is an issue I’ve been dealing with for over a decade (and before this more “recent” set of symptoms, I’d been having issues since my late teens, I’m 48 now). I had tried doing 5 tablets (chewable) in the morning and 2 more several times a day, but I have found that I have to take 10 first thing in the morning and then 10 more another 2-3 times during the day. These tablets are 25 mg of cocarboxylase, I did take the lipothiamine but have found the active form of thiamine to be most beneficial. Hopefully I can eventually cut back on the amount, but it’s been so long that I’ve had this problem that I’m guessing it may be awhile before it is resolved. I’m using the Source Naturals brand of cocarboxylase. I also am taking magnesium, PQQ, ubiquinol, biotin, s-acetyl-glutithione, and a multi vitamin mineral.
Hi Jeanette,
In the following publication Dr. Costantini reports the effects of Thiamine on 3 people with fibromyalgia.
2 of them respond to treatment only from 1500mg / 1800mg of TPP, and nothing before (like you):
“Patient numbers 2 and 3 never reported any improvement of neither fatigue nor CWP until the dose was increased to1500 mg / day, instead, an abrupt improvement occurred at doses of 1800 mg / day.
The therapy seems to be characterized by an ‘all or nothing’ effect. In other words, below average daily dose, there was no improvement observed. ”
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3669831/#R11
I have not found a cocarboxylase in chewable form, even at the Source Naturals site, only as sublingual tablets.
Dr Costantini has also succesfully treated Parkinson disease with megadose thiamine. It is becoming clear that these diseases are caused by thiamine dependency, i.e. a genetically determined defect in the binding of thiamine to its enzymes. Overcoming the defect with a megadose of the vitamin is an illustration of epigenetics, the science of how our genes are affected by nutrition and lifestyle
Thank you Xavier, I actually saw this or similar article several years and tried experimenting on myself starting with 500 mg of thiamine per day. I worked up to 1500 as I found that to be what helped. I never did more than that because I just didn’t think that more would help further as it seemed like such a huge dose. (Maybe I should have tried more) I had a level of improvement, but not like what I’ve noticed with the cocarboxylase/TPP. The tablets I’m taking are the sublingual ones you mentioned. I just put so many in my mouth at once though, that I end up chewing them a bit so they dissolve faster. They have a decent enough minty taste and thankfully are not expensive.
Thanks so much for the update, Jeanette. So glad (and encouraging) to hear you are doing much better.
I’ve been trying to understand all of this.
Can anyone tell me if the thiamine pyrophosphate supplement still needs a transporter protein in order to get inside the cell? Does it share the advantage of the disulfide derivatives?
Thiamine from diet is absorbed in the jejunum in an alkaline medium. It is then absorbed by means of a transporter and phosphorylated to make thiamine pyrophosphate, TPP (cocarboxlase). However , there is an exception. An organelle known as a peroxisome carries out an important reaction known as alpha oxidation of fats and TPP has to be imported into the organelle to bind with the action enzyme. This recent discovery means that TPP is essential for the oxidation of certain fats as well as its well known effect in carbohydrate and protein metabolism. If alpha oxidation is impaired, phytanic acid will accumulate in the blood and is another tool for discovering thiamine deficiency.
Thanks again, Dr. Lonsdale.
So if I’m understanding correctly, the TPP tablets Jeanette is taking do not need a transporter to enter the cell because TPP is a thiamine derivative which has already been incorporated into the cell and then phosphorylated, thus bypassing both transport and phosphorylation?
If that is the case, how come you do not particularly advocate its usage alongside the disulfide derivatives like lipothiamine? Does it not go straight to work in the body?
Thanks in advance for your insights.
Thanks, Dr. Lonsdale.
So if I’m understanding correctly, the TPP tablets Jeanette is taking do not need a transporter to enter the cell because TPP is a form of thiamine which has, under normal circumstances, already been incorporated into the cell and then phosphorylated, thus bypassing both transport and phosphorylation?
If that is the case, why is it that you do not particularly advocate the usage of TPP alongside the disulfide derivatives like lipothiamine? Does it not go straight to work in the body?
Thanks again in advance for any insights.
Well, that is an interesting point. TPP has to be imported into the peroxisome and it uses a TPP transporter (TptA). So Jeanette also presumably needs it because she is unable to phosphorylate thiamine
Thanks, Dr. Lonsdale.
Is the only way one knows if they cannot phosphorylate thiamine through trial and error supplementation?
Very interesting, thank you. Does the build up of phytanic acid cause symptoms? Perhaps similar to lactic acid? Maybe that’s partly what the muscle burn is from. Just a thought 🙂
Don’t worry about reducing the dose of the cocarboxylase. You have evidently found the secret of the problem and it is almost certainly genetically determined so stick with it. It is not toxic.
Thanks! I have had that in mind, just felt like I was needing such a large dose that perhaps as time went on I could taper down, but yes, I will for sure keep up with the high dose as Id rather have what my body needs and just pee out any extra, than not have enough! 🙂
Phytanic acid (PA) is derived from phytol that occurs in milk and meat. Phytol is formed as a byproduct of chlorophyl and PA is very toxic. It has recently been found that thiamine pyrophosphate (TPP) is required as cofactor for the enzyme that processes PA so finding PA in the blood or urine could be a sign of a genetic effect in the enzyme or a deficiency of TPP. PA accumulation has been reported in malignancies and can be the cause of a brain disease.
Thank you Dr. Lonsdale for further explaining this. I’m starting to wonder about something more that you may be able to help with. Before I started on the TPP, I was taking large doses of s-acetyl-glutathione. It was very helpful (although expensive), and I also started taking catalase with it as I read that catalase is needed to further break down the peroxidase that is formed when glutathione does its detoxifying. This also seemed to “feel” helpful. So, I’m wondering if TPP has any affect on the body’s production of catalase. (I had stopped taking the catalase and lowered the dose of glutathione simply because I felt I was taking a lot of supplements, and the cost does add up.) Thank you 🙂
Cocarboxylase is 18 dollars for 60 lozenges, 25 mg each.
Jeanette, you said you need 30 a day.
How are you doing now a year later?
This long and complex story is absolutely typical of what happens when energy metabolism is defective. We do not know whether this is genetic in origin but I strongly suspect that the pregnancy had much to do with it. In other words genes do not usually work on their own. Diet is of immense importance for we are conversant with the fact that alcohol and sugar precipitate thiamine deficiency. Let us look at the history. He began seizures at the age of six months, suggesting a B6 deficiency. Extreme fatigue, migraine, broken bones, sugar craving, dilated pupils, low blood pressure, POTS are all typical of an energy crisis precipitating dysautonomia. That was why he stopped growing for two years. You should look up the post on the Hormones Matter about eosinophilic esophagitis because it illustrates the diversity of symptoms that develop when brain energy is defective. The best thiamine supplement would be Lpothiamine and start with 50 mg a day. Read about paradox as written on this website and wait for the paradox to subside before you start increasing the dose. Add B complex and a multivitamin. I would be interested in the treatment that he has been given for dysautonomia because that is clearly a mark of energy deficiency in the brain.
joan Wilson says:
November 18, 2019 at 12:05 am
I am thrilled to get your answer and thoughts – it means so much. When you say the pregnancy had something to do with it, could you explain what you mean ( also, genes do not usually work on their own.). I may be a bit tired tonight, but I am struggling to understand that. As a toddler our son was addicted to sugar and he comes from a long line of alcoholics – me included. My problem was resolved 4 yrs before his birth. One set of his grandparents both were alcoholic and died from alcohol related illnesses. One grandfather on the other side had alcohol issues. Many great uncles, an uncle, brother, and sister had alcohol issues. I don’t know if this is important, but we lost 2 infants before this son; a daughter to SIDS and a son to Triasomy 18.
As our son grew we worked to reduce the sugar in his diet, but were not sugar free. From 2012 to 2014 he was on this regimen of sugary drinks and foods, recommended by a scientist and the dysautonomia symptoms ramped up from that, I think.
The neurologist handling his dysautonomia treatment has recommended compression stockings to waist, Midodrine 5 mg 3x a day, 5 mg of Propranolol 4x a day, salt intake of at least 5g per day, 2 litres of fluids, some Gatorade included, and a graded exercise program using a recumbent bike, rowing machine, swimming, or floor exercise. We are not able to do all this. He can’t drink that much, too much salt is excitatory, he cannot take Gatorade ( sugar free) or fortified foods. She advised eating every 2 hrs – he eats every 2.5 hrs and has for about 4 yrs. He cannot do the exercises often or consistently yet, but is moving more & hurting less. Midodrine helps SO much. He still cannot sit with feet down for too long. His blood pressures still stays on the low side. She also wants him in speech.language therapy, but we haven’t gotten that done yet, as he has been sick for nearly 3 weeks with a sinus infection. Oh, and he did have constant ear infections as a baby, got tubes in his ears, and then started having sinus infections.
Thank you SO very much for responding. It means a lot to u
You are cocarboxylase dependent, meaning that you NEED big doses
Hi Dr. Lonsdale,
I have severe malabsorption syndrome due to years of chronic, debilitating GI conditions. I’ve recently discovered via testing (as well as worsening of symptoms) just how deficient I am in a plethora of vitamins and minerals.
I’ve taken oral B vitamins in the past to no effect. This time around I immediately opted for transdermal/topical vitamins. I am now using a prescription thiamine hydrochloride cream (I do not tolerate Authia/TTFD due to sulfur intolerance) as well as a B complex cream from Lee Silsby Compounding Pharmacy.
My questions are these: do you believe transdermal solutions to be an effective way of receiving B vitamins, especially thiamine? And if so, how good do you think absorption rates are? How much of a dose do I need if this is my only source of thiamine/B vitamins?
I can only get the thiamine salt form at this time. Hopefully that is acceptable to build up levels in my body.
Thank you so much for your time and attention.
You can get Benfotiamine. It is a thiamine derivative whose only sulfur atom is part of the thiamine. You then do not need to get thiamine via the skin
Thank you for your reply, Dr. Lonsdale.
But what about my compromised absorption? Does benfotiamine somehow bypass gastric digestion and get straight into the bloodstream? My stomach is too damaged to properly absorb most vitamins and minerals at this point in time.
I guess what I’m asking is whether or not there is a form of thiamine best suited for those with malabsorption/enteropathy? Thanks again.
No. Lipothiamine is best in my opinion
Thanks, Dr.
I know Benfotiamine has an affect on blood sugar. Does it exclusively LOWER blood sugar? Or does it regulate?
I ask because I already have well-regulated blood sugar, which if anything tends toward the lower side, and I don’t want to give myself hypoglycemic problems should I attempt to take this form of thiamine.
Thanks very much.
Thiamine deficiency can cause a normal, high, or low blood sugar. Thiamine administration easily causes disease remission if blood sugar is normal. The response is harder if blood sugar ls high and the response is much harder if it is low. This reflects different stages of the disease as it progresses
Benfotiamine is not used to treat “blood sugar”. It is used to correct thiamine deficiency.
Hi again Dr. Lonsdale.
What sort of fasting or postprandial glucose reading do you consider to be indicative of chronically low blood sugar?
Thank you.
Dear Dr. Lonsdale
I love reading your posts on here and would much appreciate your advice.
I suffered a disc rupture (L5/s1) needing surgery 27.12.17 as it had sequestered. Ouch!! Pain relief was instant but I didn’t make a good recovery with on going burning stinging nerve pain. I used to be a heavy weekend drinker, have had high stress jobs (sister in a NICU in the UK) and life stress losing both parents before they aged 70, mum to breast cancer aged 62 and dad to general ill health resulting in an above knee amputation. In the 8 year period they passed I also had a baby. I am wondering if there’s a genetic component to my probable thiamine deficiency?
Oxalates have played a part in my poor recovery as I started keto/fasting in the year of my surgery. I now eat predominately carnivore which saw me in hospital with a T wave inversion 10 days into this diet shift.
I have been having runs of palpitations, dizziness and my T wave inversion is back which is yet to be further investigated. All my bloods are normal and I have good kidney function which is a relief as I need 7 grams of potassium in citrate/bicarbonate form for the oxalate issues. If I drop my heart symptoms/blood pressure get worse! I also take magnesium citrate 315mgs twice daily, calcium citrate 400mgs once daily.
I’ve just worked up over 5 weeks to 50mgs of allithiamine and am really struggling. Sleep disturbed again, leg/nerve pain increasing, digestion off and my heart rate was so high on a little walk today I nearly passed out. Is this paradox? I’m very concerned about the level of potassium I need and would really appreciate your words of wisdom/advice as I feel in a real mess with my health and am having a repeat ECG on Monday I just hope my t wave is normal again. I’ve not been able to work since March and feel quite desperate.
I’m convinced I have a thiamine issue and wouldn’t be surprised if my parents had issues too, sadly too late for them.
Thanks for your time
Jo
Hi Dr. Lonsdale,
I have severe malabsorption syndrome due to years of chronic, debilitating GI conditions. I’ve recently discovered via testing (as well as worsening of symptoms) just how deficient I am in a plethora of vitamins and minerals.
I’ve taken oral B vitamins in the past to no effect. This time around I immediately opted for transdermal/topical vitamins. I am now using a prescription thiamine hydrochloride cream (I do not tolerate Authia/TTFD due to sulfur intolerance) as well as a B complex cream from Lee Silsby Compounding Pharmacy.
My questions are these: do you believe transdermal solutions to be an effective way of receiving B vitamins, especially thiamine? And if so, how good do you think absorption rates are? How much of a dose do I need if this is my only source of thiamine/B vitamins?
I can only get the thiamine salt form at this time. Hopefully that is acceptable to build up levels in my body.
Thank you so much for your time.
Did you ever have success with ingesting the vitamins, rather than topical? Or did you continue with topical? My husband has never been formally diagnosed with anything except Celiac Disease, not even malabsorption, but he has all the symptoms of malabsorption. He has been taking TTFD since May. Allithiamine, then lipothiamine, and now Thiamax (he’s at 300 mg a day, currently). It’s doing bupkis. He still has Cyclic Vomiting Syndrome, still has episodes of pooping pure yellow bile, severe bloating, distension, intestines on fire, reflux, dizziness, vertigo, etc. I happened to get some 75 mg B1 patches as a sample in the mail, and his 6 day vomiting episode has finally ended and he’s starting to feel better. So I’m really wondering if he needs topical, or even B1 shots or IV.
Would he have exocrine pancreatic insufficiency or a bile duct blockage?
Hi Dr Lonsdale,
I have a 20 yr history of CFS. I have two children, and perform the least amount of paid work in order to be able to operate a household and partake in family life. Symptoms are debilitating fatigue/low BP/brain fog/exercise intolerance/lactic acid build-up/air-hunger.
I am a new-comer to B1 and recently purchased thiamine nitrate (250mg) tabs from my local health store. Iprobably took too much at first (500mg) as it put me to sleep. I halved the dose and felt increased energy, better temps, solid sleep, waking at 6am bright and fresh (as oposed the dragging myself out at 8am).
This lasted for about a week then improvements faded and cfs symptoms returned – i felt worse tbh.
From what I’ve since read, I realise this is likely paradox symptoms. I’m now on btwen 62.5-125mg (1/4 – 1/2 tab) daily, and I find I still have to take the dose at night. Daytime doses make me too tired.
I also take a Bcomplex 2xday and magnesium.
1.Are those doses ok or should I increase now despite feeling awful?,
2.what dose should I ultimately be aiming for ?
3.Would it be wise to progress to Alli or Lipo variety after finishing current supply?.
Thankyou
It is interesting that nobody ever talks about their diet on these posts. I am reasonably sure that the commonest cause of chronic fatigue syndrome (CFS) is thiamine deficiency. It may be dietetic in origin or there could be a genetic link. The very large dose of thiamine with which you started treatment would predictably cause paradox. You should maintain the relatively low dose until paradox gives way to improvement and it could last for a month or so. Once improvement occurs you can titrate the escalation of the dose to symptom relief. You also need 125 to 250 mg of a magnesium salt.
As usual, this is a frighteningly mixed up story and I am becoming increasingly aware that modern medicine makes things much worse. Few people seem to be aware that surgical intervention is extremely stressful. It easily precipitates thiamine deficiency because a surge of energy is required to meet the stress. This, together with your alcoholic binging at weekends (who does not know the thiamine deficiency connection ?) may be the beginning of your story. Oxalates are at the very end of the biochemical mechanisms for production of energy. They have no function in the body and have to be excreted. Thiamine deficiency is connected to the production of oxalates. T wave inversion has been recorded in beriberi and in fact the electrocardiogram can imitate a heart attack. However, your cardiologist will say that the person that gave you this information is either a fool or mad. Heart palpitations, dizziness and evidence of abnormal potassium metabolism are all typical of thiamine deficiency. It sounds to me as though your introduction of thiamine recently is an example of extremely severe paradox. You are taking colossal doses of supplementary nutrients and I cannot tell you how much this interferes with the whole process. The body is an intricate biochemical machine and its logic is very precise. This type of approach needs research. The problem is that the medical profession has totally rejected that nutrients of this nature are the key. Energy production and its consumption are the keys to health. The ancient Chinese wisdom of Yin and Yang (moderation in all things) is the philosophical prescription for health.
Dear Dr. Lonsdale
I so appreciate you taking the time to respond. Whenever I try to reduce the potassium, or “dump” oxalates my blood pressure rises and I feel dreadful so I feel stuck until the thiamine hopefully starts to correct things? Is this likely? Would you advice a reduction? Should I balance it out with a bit more magnesium temporarily? I really don’t know which way to turn as my doctor has no idea.
Yes they did say my ECG could have been an inferior infarction but thankfully cardiac enzymes were normal. I’ve not yet seen a cardiologist and don’t really want to as they will no doubt want to prescribe more chemicals in the form of medication!!!
I have stopped drinking alcohol since March and eat good fats and protein. I have also stopped coffee as I used to have 4 to 5 cups a day.
Any advice on how to wean my minerals would be greatly received.
Kindest regards.
Jo
Dear Dr. Lonsdale
Well my T wave inversion has gone which is good but my paradox continues unfortunately with insomnia, palpitations, shivers, panic and anxiety and high blood pressure so I’ve cut back to 25mgs allithiamine.
Do you have any suggestions on what mineral replacement you would recommend for someone in my situation as I understand the colossal doses advised are not the norm and it deeply concerns me so I would like to safely reduce.. My bloods markers were all fine today. Potassium 4.1 and e gfr >90 which is a relief.
Thanks for your time. I feel relieved to have found this page.
You may be surprised to know that you have classical beriberi. Fatigue, low blood pressure, brain fog, air hunger and above all, lactic acidosis are indeed the symptoms. Although the commonest cause is dietary, there is often a genetic background that explains why megadoses of the vitamin have to be used. The very high dose with which you started was predictably a cause of paradox and this can last for a month or so before improvement begins. Maintain the relatively low dose until that improvement begins and then you can titrate the dose to the alleviation of symptoms
Thankyou for your reply Dr Lonsdale. Your advice is very much appreciated. I have to be honest here and say that I didn’t mention diet because my diet of the last 12 months contains considerably more sugar than it has in my whole life! Natural sugars though ie fruits/honey/oj, Dr Ray Peat style, as you’ve probably guessed.
It was time to try something new I felt , other than sugar-free/dairy-free that I’ve done for 20 odd years. I will always remain gluten free.
I’m hoping with the addition of B1, my metabolism skyrockets to a whole new level.
For almost 20 years I had tingling and fatigue problems.
I’d try to exercise, but then would have to sleep (really, pass out) immediately afterward. Many afternoons I’d have a “slowdown” where my brain would shut down, literally putting me into a stupor or sleep state for 30 or 45 minutes, until my brain came back online again. During these years, I lived a normal and active life, but it was very hard! Everything felt like a struggle. Normal activity felt like rolling a boulder uphill every day.
My feet would always tingle, my hands and face would occasionally tingle. I also had tingle “waves” that would pass over my skin, across my whole body.
A neurologist ran tests (MRI, nerve conduction) and said I didn’t have MS or a brain tumor. My blood tests were normal.
I researched my problem continuously, trying supplements and elimination diets. I found things that helped: GFCF diet, Ceylon cinnamon, raising my vit D and magnesium levels, sublingual B12, methylfolate (could dampen tingles if I took a super high dose), potassium chloride. I tried literally dozens of things.
Finally, in the Low Oxalates group protocol (one of my experiments was reducing oxalates), I discovered allithiamine. Magic! When I took an allithiamine capsule, the fatigue and brain fog lifted for a short time. I took two capsules several times a day, and always took some before meetings when I needed to think clearly. I would also take one whenever a slowdown hit, and it would bring my brain back in minutes. Eventually I was able to go to one capsule a day, and then just occasional use. So, allithiamine helped my overall fatigue and brain fog, was life-changing, but I still had tingling and post-exertional fatigue.
Then I read one website that said patients experiencing neurological symptoms should try B12 injections even if their blood tests are in the normal range (mine were). I found a doctor who offered B12 shots for $35 in his clinic, and that was it! The answer. Now I self-inject 1mg hydroxocobalamin weekly and it keeps me healthy. Over the past year my tingling has faded, and now it mostly bothers me when I run (now I can run!!!!).
I also take a large number of supplements daily, including allithiamine, methylfolate, and B-complex.
A remarkable story. Don’t hesitate to push the dose of Allithiamine. It is non toxic. Also remember magnesium. It is essential with thiamine.
Thank you for reading my story. I’m very grateful for the online information you have provided about thiamine over the years. You’ve helped me and so many other people recover from illness.
I think I’ll increase my daily allithiamine from one capsule to two for a while, to build up my thiamine stores. My sibling was recently diagnosed with Parkinson’s (PD), and some people with PD are using thiamine to reduce their symptoms. Hopefully keeping my level high might reduce my risk of developing PD.
re: magnesium, I take three Jigsaw magnesium tablets per day (375mg elemental Mg), along with a large number of other supplements.
I would love to know what caused my health problems, but my tests have mostly been normal except for Graves’ disease. Even my genome looks okay, with no problems on the thiamine or cobalamin transporter genes. It makes me a bit uneasy to keep my B12 level so high, so I’d really like to understand why I need it.
Have you seen others who needed high doses of both thiamine and B12 to recover?
I feel a need to make further comment. Notice that she describes “passing out after exercise” and sleeping for 30- 40 minutes at a time. She describes her fatigue as “like rolling a boulder uphill” a very good analogy for the kind of knockdown fatigue experienced by so many people. She states that blood tests and genetic testing were all normal, leaving the possibility that dietary deficiency is the underlying cause. She found that Allithiamine “was magic” and finds that she takes it before meetings where “thinking clearly” is required. Note that she asks whether you can “build up thiamine stores” in the body and the answer to that is— no, there is no storage mechanism for thiamine, meaning that daily diet is responsible for obtaining it. It is interesting that she has a sibling who has Parkinson’s disease and a doctor in Italy is now treating this disease with large doses of thiamine given by injection. She also states that she has Graves’ disease. Thiamine deficiency causes the disease called beriberi, a Chinese word interpreted as “I can’t, I can’t” The symptoms can imitate virtually any disease described in medicine. For example persistent vomiting and abdominal pain would be unlikely to cause someone to think of the brain as causative, but energy deficiency affecting the hind brain causes the individual to be maladapted, meaning that the unfortunate patient is wide open to any form of environmental stress, including any infecting organism. Evidence is accumulating that cellular energy deficiency is the cause of cancer and if this is true we should certainly begin to respect nutrition much more.
I’m having trouble finding a source for alithiamine that doesn’t have bad additives.
Try Thiamax. It’s additive free. I comes in a higher dose though.
Your symptoms sound very similar to mine. Thiamine (in several forms, including cocarboxylase, allithiamine, etc.) and magnesium, biotin and magnesium massively improve my symptoms.
Hi there I have autonomic dysregulation of blood pressure resulting in low blood pressure I have an MTHFr gene Marriott with only moderate activity and have low B9 and B 12 I have supplemented those with no problem I went to add Benfotiamine 80 mg twice a day I got a very bad headache and even lower blood pressure by more than 20 points I’ve read this is a common side effect have you had any experience with that and is it something that will get better once you get used to it
Low blood pressure is typical of beriberi. Your headache and further drop in blood pressure after taking benfotiamine is typical of what we have called paradox. You can read about paradox on Hormones Matter. I have no idea why your pyruvate and lactic acid were normal if you have thiamine deficiency but the paradoxical reaction you got from taking benfo does suggest that it is an important supplement. I would suggest that you restart the thiamine at a much lower dose, perhaps using thiamine hydrochloride at 100 mg a day, together with 125 to 250 mg of magnesium salt. You could also try Epsom salts baths that would give you magnesium. You have to continue experiencing paradox until it begins to give place to improvement. People usually make the mistake of interpreting paradox as “side effects”, but the mechanism is completely different from that of pharmaceuticals.
Hello Dr. Lonsdale, I just took 5 days of Metronidazole combined with Augmentin for an abscessed tooth. I quit the course because I felt horrible on them, and found metro girl blog and read about Metronidazole there. I want to begin thiamine supplementation, and I just found lipothiamine on Amazon, by Cardiovascular something, now I can’t fully remember. I also read about the necessity of taking magnesium along with the thiamine. The lipothiamine I mentioned above contains magnesium stearate. Is that magnesium adequate? Or do I need to get a separate magnesium supplement? Also I have IBS and hope my gut can tolerate all this. Thanks so much for your help!
Cardiovascular Research and Ecological Formulas are two names for the vendor of Lipothiamine and Allithiamine, both are thiamine tetrahydrofurfuryl disulfide, but Lipo is enteric coated to get through the stomach acid to the jejunum for maximum absorption. Lipo does not contain magnesium but does contain a small dose of alpha lipoic acid that assists thiamine. You will need comparable doses of 250 mg magnesium tartrate. This combination will help your IBS, but beware of paradox.
Is it magnesium tartrate or taurate that should be taken with thiamine. I’m not finding tartrate for sale. Thanks!
Taurate is the word. I am sorry—-tartrate was a mistake and doesn’t exist!!!
Ok, thank you, that explains it! I currently have mag citrate and mag glycinate, but I also have taurine. Would it be just as effective to take some taurine with my mag supplements? Also, would it be helpful or harmful to take Ribose, would that be stressful to the body if thiamine is depleted since ribose is a sugar? Thank you!!
Nutrients never stress the body. Ribose is a “building block”
My lipothiamine arrived so I will be starting that. For the magnesium you mentioned using comparable doses of 250 mg magnesium taurate. Erin on the metro girl blog said that some people had found that Magnesium L-Threonate had been less stressful for their guts, so I ordered some and started it before I saw your answer to my original post. At that point the only B1 I could find in town was thiamin hcl. My gut has been a wreck so I’m hoping switching to the lipo will help. As far as the magnesium, is your suggestion of the magnesium taurate better than the L-Threonate? Are they the same thing, or enough similar? Thanks so much for your help!
They are all magnesium salts and their chief effect is to deliver magnesium. Their major difference is only their capacity for absorption, with the exception of Mg taurate. Taurine is known to have a biochemical effect in its own right, so its coupling with Mg is an advantage but not a necessity.
Thank you for all the information, and for all the helpful specifics, Dr. Lonsdale. I’m so, so grateful!!!
And in case of paradox I should just decrease my dosages, is that right? Thanks.
No, maintain the same dose until paradox gives way to improvement.
This is such helpful information! Thank you for sharing this. I’d not known of Allithiame or lipothiamine before this. I’ve been very suspicious of some kind of issue with thiamine absorption, depletion, or lack of conversion to active form, etc in me for some time now. I have had help with energy and mental clarity when taking 1500mg and up of regular thiamine, but I still struggle with debilitating muscle burn/fatigue with even just walking and a feeling of lack of blood flow when being still. I also experience chronic migraines, this past summer I had one that lasted 5-6 weeks along with what I can only call “mental stupor”. I am awaiting a purchase of a bottle of “active B1” (TPP, cocarboxylase) and am wondering if that form has much merit. (I am going to purchase the Lipothiamine now, so will be able to take both) Is it worth continuing with the active B1 form, or are the lipothiamine and Allithiamine superior?
Lipothiamine and Allithiamine are different versions of the same thiamine derivative and are superior to water soluble thiamine salts and cocarboxylase. No harm in adding them but unnecessary. You should get benefit from migraine.
Thanks! I received my carboxylase yesterday and took 10 tablets (25 mg each) and had increase in mental energy and clarity within an hour so I took 5 more a couple of hours later with good results. I took 10 again this a.m. and am maintaining better mental clarity and energy and I think even a bit of a decline of the muscle burn. I plan to take another 10 shortly. Once the lipothiamine gets here I’ll add that and most likely back off on the carboxylase. Is there a known condition where the body is unable to convert inactive to active, perhaps even from having had a virus or vaccine? (I have EBV) I also have had much better results with methylcobalamin as opposed to cyano. I think what you had mentioned somewhere about epigenetics is spot on. Thank you again for your reply and all the helpful info, it’s greatly appreciated and needed.
Interesting question. Cocarboxylase is thiamine pyrophosphate. Two molecules of phosphate are added to activate the vitamin in the body.
Thank you! By what process are the phosphate molecules added? I have gotten your book co-authored with Dr. Marrs, so if this process is explained in the book, you could just direct me to which chapter. I have the Liipothiamine now and have been taking that, but I am finding that it is the cocarboxalase that is giving me the pronounced improvement. Yesterday I had a migraine melt away after taking 5 tablets. I’ve run out (have more coming tomorrow) and now the migraine is back. I read in your book that TPP is not stored in the body, so if I do perhaps have some issue with conversion, I’m guessing it’s something I will need to take throughout the day.. I have literally been crippled by this issue over the years dealing with extreme fatigue, muscle pain/burn with exercise, peripheral neuropathy, balance issues, POTS, gastrointestinal issues, trouble with working memory and brain “fog”, dysphagia, arrhythmias (PAC’s, PVC’s, SVT) and chronic migraines. I’ve had the diagnoses of CFS, MS, and POTS. My “gut” tells me, and your research all but confirms, that my true issue is thiamine deficiency of some form. I am so grateful for this research and it being made available to the public like this. Are there any genetic conditions that inhibit the TPP conversion process? I also have a sister with Fibromialgia who has benefited from high dose thiamine (she also has had EBV in the past, but I believe her titers all went back to normal unlike mine). Your thoughts are very appreciated.
It sounds like the biochemical lesion is the phosphate addition because cocarboxylase is thiamine pyrophosphate. Since that appears to help you I would focus on that.
Thank you, I’m working on figuring out how much of the cocarboxylase I need. I’m still taking Lipothiamine as well, as I have it and figure it won’t hurt. Of possible interest, my parent’s first baby died from SIDS (not that that means he definitely had a thiamine deficiency) at one month, just makes me wonder if there is some sort of genetic. Just interesting to me.
SIDS is caused by a defect or a dietary deficiency in thiamine metabolism. I have seriously considered an inability to phosphorylate thiamine, so cocarboxylase make possible sense for you
Thanks for your input! I’ve been doing 150 mg of Lipothiamine each day and 5 tablets of TPP each morning, then 2 more every few hours . I can feel things changing for the better in me although there’s still a ways to go, I’m very encouraged by this and again, am so grateful to have this validating information.
Hi Jeanette,
I was wondering where it was you purchased your TPP tablets from? I’m very interested in trying this form of thiamine as I have awful sulfur sensitivity.
How are you getting on these days?
Thanks!
I’m 28 and it has been several years since I could recall a dream. On day one with Lipothiamine I could easily recall my dreams. This didn’t happen with benfothiamine, and now I understand why. Your post Derrick is life saving. Thank you 🙂
Interesting. Thanks for the comment
That is very interesting. The point of all of the information in these posts is really very simple—–energy! I nave come to the conclusion that disease is nothing more than ENERGY DEFICIENCY ! Of course, thiamine is not the only catalyst that enables us to use oxygen in the process of creating energy, but it is at the very gate of entry of glucose into the chemical “cellular engine” known as the citric acid ( Krebs) cycle. This is not a new idea: a researcher by the name of Hans Selye stated that human disease was simply “the diseases of adaptation”. He formulated the complex way in which a physically stressed animal adapts to the STRESS imposed by trauma applied by the researcher. He called it “The General Adaptation Syndrome” (GAS) and was able to show that the lab studies during the process of adapting were the same as the lab studies found in sick humans. In other words the humans are going through the GAS that is the manifestation of their illness. Selye indicated that the GAS was energy dependent and one of his students was able to show that thiamine deficiency could result in an animal developing the GAS. I visited Selye in Montreal in 1973 and he indicated to me that his work would drive the medicine of the 21st century. Unfortunately, his work is thought of as interesting history but its application to human disease has never been used to date.
Do you have suggestions on transdermal thiamine? I haven’t been able to tolerate any thiamine or supplements for many reasons including great autonomic dysfunction and a very unhappy gut. I’ve recently found transdermal patches but the company I’ve been using groups vitamins/minerals/antioxidants/ingredients together and they do not carry patch singles but I have seen thiamine patches on the market as a mosquito repellent which is very interesting and would love to hear a post on that. But would like suggestions on a transdermal option do deal with the multitude of symptoms; the most pressing being debilitating fatigue, mitochondrial and autonomic dysfunction, blood sugar dysregulation, etc. Much appreciate and thank you so much for your articles and vital info!!
There is a transdermal cream called Authia . Try Ecological Formulas for info
Dr. Lonsdale,
Any thoughts on thiamine supplementation in an older adult on dialysis?
Unidentifiable micro bleeds in bowels are being attributed to the cause of dropping Hgb and subsequent blood transfusions. I think 13 L transfused in one week recently.
I’m hopeful that thiamine will help, concerned about dosage/dialysis.
Dialysis 2x/week 80 year old male history of T2DM.
somehow my comment was not posted–so, repeat
fast losing any nerve sensitivity in legs, brain slow, ataxia–also had optic neuritis years ago, now again and with glaucoma–
have tried thiamine HCL with no effect
then read Dr Lonsdale and Chandler Marrs info–got Allithiamine, severe itching all over, similar with Benfotiamine–but benefits so good, am continuing these supplements despite the itching–am also just trying sulbutiamine, so far, no bad effects–
what could this severe itching be caused by, and is it detrimental to continue despite it??
an very grateful for the life-changing info from Dr Lonsdale and Chandler Marrs–
thank you
Probably “paradox” that may last as long as a month or so. Stick with it.
Is it advisable to build up slowly to help mitigate some of the paradoxical symptoms?
Your work here is amazing.
Thank you.
I did stick with it, and got thru almost complete loss of sensation in feet and legs, to now back to walking several miles per day!! a painful, but relatively quick process considering how very long this decline has been going on–still some problems, but SO much better (also do need much B12, shots and sublingual)–
one point of interest to you Dr Lonsdale–
your message must be getting out there, because the price of Allitihamine on amazon went up by 25% just recently 🙂
Hi e, Look up B6 toxicity – I read that B6 can deplete B1, and conversely if you were B1 deficient I suspect you could not balance the abundant B6 we get from food. Your symptoms match, and the paradox effect makes sense as it is the nerves healing.
Hi, just a thought about the severe itching you are having with those supplements…I have experienced a severe sensitivity to magnesium stearate which at one time caused itchy bumps all over me, I went to the dermatologist and had biopsy which showed a reaction to an environmental agent, so through process of elimination I discovered it was to the additive magnesium stearate which is used as a flowing agent in many supplements (it’s also called vegetable stearate). It’s very possible you are having the same reaction. I try my best to avoid pills with this in it, but it can be difficult. I often end up buying bulk pure powders and making my own capsules.
Maybe something to do with inability to metabolize sulphur?
Dr Londsdale,
I am thiamine and magnesium deficient. I have been taking thiamine hcl and magnesium liquid and slowly recovering from stress, foot and wrist drop, muscle atrophy, heart palpitations and general well being.
I have tried thiamine derivatives before and reactions are not good. Happy with hcl but slow progress. I have also been taking b5 for adrenal support.
Is the protocol I am following good ? Is thiamine hcl effective?
Thanks.
F
If you get relief, it works
This is really interesting. Five years ago I went through a very traumatic event. My whole family did. We all had different symptoms. Mine was the worst. One day I just stopped going to the bathroom, anxiety, hypervigilance, mood swings, etc. I saw three gastro docs. They claimed I wasn’t following their directions and that I should come back in 6 months. I ended up in the ER. So I decided to take things into my own hands. I took classes in biology and also researched on my own. It took 2 years but I have healed most of my symptoms. I thought choline/inositol was the answer, it was helping a lot. but soon realized in my research that it was just forcing the acetylcholine. Not fixing what was wrong. The B1 has been a godsend and I believe my adrenals are working much better. I use to pass out when stressed and now I can recover better. My husband says he has more inner peace and his tremor is much better. We thought he had Parkinson’s. As a family, our symptoms were all over the map. I still have cognitive issues but have switched to allithiamine now. One cool symptom was that my eyesight is 80 percent better. I thought I was going blind. Also, I am not constipated and my muscles are tighter, hard to explain but all over my muscles have firmed. I think my boobs are getting bigger. My hormones must be working better. I had all the symptoms and I thank you for your research.
It sounds like inherited mitochondrial disease. You probably know by now that mitochondria have their own genes and they are passed to children by the mother only. Dad’s Mt genes are lost at inception. Thiamine seems to be a driving force even for this inherited situation because it is the prime mover in producing cellular energy. The citric acid cycle is necessary for producing acetyl choline, hence the importance of thiamine. Allithiamine or Lipothiamine are your best bet.
Hi Dr. Lonsdale,
I’v not been taking Lipothiamine for a little over a month. I believe the main symptoms of “paradox” have resolved which were emotional volatility, extra physical anxiety and chest pains. I have now increased the dosage to 200mg lipothiamine per day split in the following manner: 100mg at breakfast, 50mg at lunch, 50mg at dinner.
Note – I have not yet felt benefit from this regimen for my longstanding Fluoroqunolone antibiotic toxicity characterized by peripheral neuropathy and musculoskeletal pain or longstanding anxiety/OCD/insomnia.
I have noticed that, since increasing Lipothiamine, my nasal cycle has become more balanced and the feeling of one nostril totally plugged has receded to a further degree. This is especially true when I expend energy, like taking a walk. The response is unmistakable. I should also like to add that I speak with a stutter, and I have read that thiamine may help this condition. I have not noticed improvement in this area yet.
I do wish to get your book, and will do so when my budget allows.
Here are further questions if you would like to entertain them.
1. Is there a “mini” paradox associated with the raising of the dose? Perhaps I notice 1-2 days of such symptoms when I have increased.
2. You previously advised to raise the dose “gradually” until symptoms yield. Do you ahve a general schedule to increase? For example, I waited at 150mg for a week, then I raised to 200mg.
3. Since you indicate that TTFD is better absorbed that Thiamine HCL, I am assuming that smaller doses of TTFD are more effective that Thiamine HCL. Is there any general dose equivalent between these substances?
Thank you for your time and attention.
Geez! I found yet ANOTHER symptom of thiamine deficiency on this site as I am gobbling up this info like crazy now! I also have mitral valve prolapse which is a thiamine deficiency symptom/disease. So, I tried to search this out online but I also had hyperparathyroidism (parathyroid tumor). Is thiamine also involved in calcium metabolism? (plus, I asked earlier about Marfan Syndrome). I am 65 years old and have suffered a lot of health problems for a long time. I have seen so many doctors and sought help and only got blank looks for so long. This site is SUCH a relief!
Mitral valve prolapse (MVP) is a very interesting modern phenomenon. Many years ago, a smart cardiologist recognized that it was associated with problems in the “brain computer”. This is the part of the brain that organizes and controls the autonomic (automatic) nervous system. The “brain computer” is very sensitive to thiamine deficiency but the connection with thiamine has not yet clicked in medical literature. Interestingly, a paper showed magnesium deficiency in pediatric MVP and Mg treatment ” relieved chest pain” but did not help other symptoms. The authors evidently were not aware that magnesium and thiamine work together in energy metabolism and that adding thiamine would have resolved the other symptoms. MVP is due to myxomatous degeneration of the valve and is evidence of connective tissue problems so that Marfan’s syndrome or Ehrlers Danlos syndrome might occur concurrenly with MVP. Everything is connected to everything else!!
Hi Dr. Lonsdale;
I just found an old online post of yours that kind of stunned me. I don’t know if I misunderstood what I read or not so I thought I’d ask you. One of my “weird” symptoms is that I get very confused, off-balance and a bit anxious if I go out into bright sunlight. I just read a post of yours where you mentioned some workers who ate their lunch outside and were fine until sunlight hit some of them and they displayed symptoms of beri-beri. I have been trying to figure out what the heck this thing is with me and going out into bright sunlight. Did I misunderstand what you were talking about or is this another beri-beri symptom? I am stunned #1 because I found this post accidently and #2 because you are so full of incredible information.
Interesting question. The factories in Japan were built in blocks and the workers would take their lunch in the corridors between them in the summer. Initially in shade, as the sun came round and shone in the corridor, a group of workers would get their first symptoms of beriberi. It was natural for the investigators of that time to conclude that beriberi was due to a mysterious infection before the discovery of thiamine, the “anti-beriberi factor” that had been later discovered in rice polishings”.Now we know that thiamine deficiency is the cause of beriberi, what is the explanation??
Sunlight stresses the human body and automatically initiates an energy requiring adaptive whole body response, organized and conducted by the brain. For the workers who were in a pre-lunch marginal state of malnutrition, the exposure to sunlight initiated a stress response and their brains could not muster the extra energy requirement to meet the demand. Thus, the symptom that you describe shows that your brain is “weighed in in the balance and found wanting”, like the Japanese workers. The reason for this kind of “mystery” in America is because of our hedonistic attachment to sweet tasting things and alcohol. Also, the highest IQs are they. greatest risk because those brains have a higher rate of metabolism and require more energy.
It is amazing to me to have an answer to a symptom so weird that I doubted myself that it was actually happening. I thought I had to be imagining it! Thank you so very much! If I had not seen that post I very well could have never had an answer or any validation for that matter! And especially thank you for the last sentence of your answer where you implied I am a genius! I think you may be right! : ) I ordered that transketolase test from London a couple of weeks ago. I had it all figured out up until the couriers could not guarantee it would get there on time without getting stuck in customs. I had steroid poisoning a couple of years ago and it took me 9 months to wean off of them. I think that made things much worse. I also am heterozygous for the MTHFR gene. I assume I have some kind of thiamine issue too. Dr. Lonsdale, we have Marfan Syndrome in our family, having claimed my grandfather, mother and my little brother. Is there some way thiamine/beri-beri could be a factor here too? I know it’s an out-of-left-field question and I know it is a collagen problem but I am wondering how much B-vitamin issues have made my family sick? Thank you!
Marfan syndrome is caused by a genetic mutation in a protein that has to go through a complex folding of its amino acid chain to become functional. It causes production of a substance called fibrillin, essential to the construction of connective tissue. Although 75% of cases are inherited, in another 25% the mutation is spontaneous. The interesting question has been asked as to whether Marfan syndrome can be related to thiamine deficiency. The effects of this disease get worse with aging, implying that energy metabolism gets into the act. I have just published a paper in Medical Hypotheses, proposing that thiamine initiates the energy production that enables the complex protein folding mechanism to occur
Thank you!
I don’t know if this is related, but I had a very thiamine deficient child who never, ever tanned, no matter how long he was outdoors for (he didn’t like being outdoors much). If he was in the sun for too long, he went straight from pale to burnt. I started giving him thiamine after nutritional testing suggested deficiency, and he developed a pale tan literally overnight. After that, he continued to tan normally in response to sun exposure.
Thank you Dr. Lonsdale for your response to my question.
Thanks for your reply, Dr. Lonsdale.
I dropped down to 50mg for a few days. Then back to 100mg. I can report that the more balanced nostril breathing continues to arise several hours after each dose of Lipothiamine. It returns to dysfunction afterwards.
I tried to go up to 150mg a few days ago – with the last 50mg dose at dinner – but that seemed to correlate with an increase in anxiety and insomnia. I cannot say whether this connection is real, as it could be the normal vicissitudes of my anxiety – but it has been severe.
I do not drink alcohol or have large intakes of sugar, but will be mindful of sugar intake.
Currently consuming 100mg Lipothiamine. I do not yet see improvement with neuropathy, musculoskeletal pain, insomnia or anxiety with this, but I only added the Thiamine on June 18.
I have a few more questions for you:
– Should one refrain from taking TTFD supplements in the evening. Are they known to interfere with sleep?
– Is it okay to take 100mg at one shot with a meal?
– Is the paradox prone to restart when one increases the dose?
Thanks again.
Wait out paradox
All very interesting! You are a good “guinea pig”. 1. TTFD is usually best taken in the morning for a one a day dose 2. It is OK to take 100 mg, usually as a daily dose. 3. paradox is caused by shifting from a catabolic to anabolic state i.e nitrogen loss to nitrogen gain. Your guidance system is the nasal cycle. The neurological benefits will come much later. I conclude hat you have a genetic risk factor, possibly a missing thiamine transporter. I was interested in the fact that the low dose gave normal nasal breathing for only a short while and wondered whether a repeated low dose would work better for you than a single daily dose. I must remind you that this is true pioneering. It is hard enough to treat long term nutritional deficiency without a genetic background, but it has seemed to me that a surprisingly common genetic defect that accelerates thiamine deficiency is in one or more of the missing transporters. We are beginning to realize that polymorphisms (look this up) are common and do not trouble us until another factor such as nutritional deficiency creates clinical manifestations. The new science of epigenetics is a very important addition to genetics. The trouble with all this is how you get the scientific knowledge to the suffering patient. This is what this website is dedicated to.
Hi Dr. Lonsdale,
Thanks very much for your continued reply. Can you please outline from your above comment what “a repeated low dose” would look like in terms of scheduling on a daily basis? If the total dose is 150mg, would it be two 50mg pills at breakfast, followed by one at lunch?
Regarding genertic reisk factors and a missing thiamine transporter – I have my raw genetic data available through 23andme. I have not been able to locate the SNP and genotype combination to affirm this suspicion. I have researched this to no avail. Do you know if there is a specific snp/genotype I should check? If so, I can report back.
The improved nasal cycle breathing continues some hours after each dose of thiamine. It is difficult for me to accept that this is from thiamine administration, but the effect is unmistakable and has altered how my nasal breathing has been for my life. I am still on 100mg of Lipothiamine and plan to increase to 150mg tomorrow if I can tolerate it.
Thank you for your time
Try 50 mg 3x/day instead of one dose of 150 mg Lipothiamine
Is it a bad idea to take that last dose of Lipothiamine at dinner or later in the evening? Could it cause insomnia?
Thanks for your time.
One daily dose of thiamine or one of its derivatives is all that is necessary. Although it is a vitamin, not a drug, it is being used as a drug when given in megadose. It requires a view that is completely different from present day conventional medicine, so people that want to take advantage of its benefits need to buy our book and skip the technical sections.
Hi Dr. Lonsdale,
I’ve been taking the Lipothiamine this week. I’ve altered between 100mg and 50mg per day with meals. Sometimes the 100mg dose is too powerful. I think the effect is too energetic. I noticed that I get increased chest pains and racing thoughts/anxiety with ingestion of Lipothiamine. Also, my insomnia has been very bad. I can’t tell if all of this is normal waxing and waning or if this is the “paradox” you discussed.
Yesterday, I noticed that I was able to breath through both of my nostrils for a period of over three hours. This is highly unusual, as I have a deviated septum and one nostril is always very, very clogged. Then I saw your paper that discussed “Exaggerated Autonomic Asymmetry.” I wonder if this change in my breathing was chance coincidence or validation of the function of Lipothiamine. My breathing has since mostly returned to dominant nostril breathing.
You are indeed experiencing paradox. Continue the lower dose. The nostril problem is because of a sick nasal cycle, a function of the autonomic nervous system, whose controls in the brain are highly susceptible to thiamine deficiency. Stop sugar and alcohol, both of which induce thiamine deficiency
I’m sorry my first comment didn’t post so I must have made a mistake.
Briefly, I am a 59yo, male Type 2 diabetic of 20yrs. About 8 years ago I began to develop what I think now was B3 deficieny. It was a dark area around my neck (Casal’s necklace?) and by its characteristics it did not seem to be from sun exposure. Anyway, at the time I thought something was deeply wrong with me besides, or because of my diabetes.
I began to pay more attention to my nutrition and supplements which included the water soluable vitamins, but I was not consistent with either.
It was later on when I was reading, really by accident, an article about Benfotiamine and the topic of B1 and the dry form of Beri Beri that the light went on for me. I had had leg weakness, the beginning of brain fog, neuropathy, and my bowel had basically stopped for a period of time.
I have been trying to build up my Mg for a year. I take B6 with it. I am getting the multivitamin, and I get around 200mg B1 salts?a day. I have taken Benfotiamine from time to time but I tend to return to commonly available B1.
I realize now, no sugar.
From reading the other posts here, I wonder should I switch to Lipothiamine because I still have the brain fog? But I don’t know if it the dose is 1:1. In other words, if I was taking 200mg of the salts form, should I work up to 200-300mg (LI)? Thank you. I had posted the study regarding diabetic kidney disease, sorry this is out of order.
The major clue is diabetes. Both types 1 and 2 are reportedly thiamine deficient. There is genetic risk for both. Thiamine needs a genetically determined protein transporter to pass into cells and absence of one or more of them seems to be quite common as a cause of thiamine deficiency. I suggest Lipothiamine 100 mg/day (Ecological Formulas) + magnesium taurate 250 mg + well rounded multivitamin.
In my earlier comment this was the study I was referring to. Univ. of Warwick. ”
Vitamin B1 Could Reverse Early-stage Kidney Disease In Diabetes Patients”. https://www.sciencedaily.com/releases/2008/12/081208092149.htm
Yes, I am familiar with Thornalley’s work
Hi Dr Lonsdale,
In what cases , if any, might you suggest someone have intravenous or intramuscular thiamine?
Would this give quicker results? Would it bypass paradox symptoms?
If it’s hard to come by or needs a script I’d probably sooner stick with tablet form- I’m a bit over dealing with GP’s.
But if you can get it online and self-inject, that could be an option.
Hi Kirsty – I see you’ve yet to get a reply from the Dr. – I do wish this site does allow for reply notifications…
The IM injection will not bypass the paradox symptoms. It merely increases the absorption rate around 140 times higher than oral dosage. Hope that helps.
I am a 59 yo male, Type 2 diabetic for 20 years. I noticed about 8 years ago what, looking back on it, may have been the beginning of Casal’s necklass (B3) around my neck. Of course, I did not know the symptoms of B3 deficiency at the time, but I felt intuitively it was not just sun exposure which was causing the darkening. Did I have early symptoms of Pellegra? I don’t know, I am not a doctor.
I knew enough about the water soluble vitamins to know I needed them. I knew I urinated more than normal and so on. So I was taking vitamins and minerals but I was not entirely consistent in use.
The light went off for me when I was reading about Benfotiamine and the author listed the symptoms of Dry Beriberi. I was stunned. I cried. I got serious about nutrition, vitamins and minerals. This led me further into studying B1, and now to Dr. Lonsdale and his very important work.
As I understand from above. I have neuropathy but it much improved and I am much more functional now that I make sure U get the thinks a diabetic needs. Replenish.
Do I nderstand from the above that Allithamine may be the best form of B1 for me? I have been working to increase my Mg appropriately for a year now. I also take appropriate supplement etc. multivitamin.
Second question, might B1 also slow down the progression of kidney disease or help prevent it. I read a study, sorry I can’t find it, that 300mg/day, divided, of B1 salts? (I’m not sure what form was used in the study) may slow progression of kidney disease? I realize this is probably a therapeutic dose and should be under supervision of a doctor. Thank you.
See above.
I have just started exploring the benefits of Thiamine. I am wondering if it is something that a person with Autoimmune Autonomic Ganglionopathy. (AAG). M brother has just been diagnosed with this autoimmune disease. He, of course, has gastorpersis, low blood pressure with syncope, dry moth, low heat tolerance, brain fog, and just a host of other issues related to the disfunction of the Autonomic Nervous System. If his insurance approves it, he will start IVIG infusions. Can adding the Thiamaine supplement help him at least with the fatigue and brain fog?
I have been interested in the connection between thiamine deficiency and autoimmune disease for some time. What I do recognize in your brother is the disaster of energy deficiency. His symptoms are those that occur in the classic disease beriberi, the prototype for dysautonomia. I do not understand the rationale of IVIG. I have seen many patients with this misdiagnosis and there is no doubt that it is extremely common because of our love of sugar and alcohol, both of which induce thiamine deficiency. He should respond to megadoses of thiamine but you should be aware of paradox, also known as “refeeding syndrome”, that is discussed on Hormones Matter and it should be under the care of a physician. However, I am afraid that your physician will openly scoff at this. He/she will tell you that because of vitamin enrichment by the food industry, vitamin deficiency in America simply does not occur and that your informant is an ignorant trouble maker. It is very sad for me to witness so many medical disasters. The medical literature is full of thiamine responsive cases but the papers are from countries other than America.
Hi,
Is there concise information on which thamine supplement to use and what dose? I have read through several articles by Dr. Lonsdale on this website and their comments, and a thiamine paper on pubmed. I have gleaned the following:
1. Regular “thiamine” at 1500mg to 1800mg may have the potential to help certain pain conditions.
2. Allthiamine and lipthiamine are a superior form as they cross the blood-brain barrier (BBB).
3. Benfothiamine may help peripheral tissue but does not cross the BBB.
What is the proper dose of allthiamine/lipothiamine and is that dose equivalent to the 1500mg regular thiamine that may help pain conditions?
Primarily, I am looking to treat peripheral neuropathy and diverse musculoskeletal weakness secondary to a toxic reaction to a fluoroquinolone antibiotic. My neuropathy has been unresponsive to benfothiamine at 450mg, magnesium glycinate/malate at 800mg, R-ALA at 300-600mg, DHA fish oil at 3G, PQQ, B12, Vit D, and several others.
Thanks for your time.
I would suggest that you try Lipothiamine from Ecological Formulas. Start at 100 m g and increase gradually depending on response. Take with 2-3 hundred mg magnesium taurate and 1.2 hundred mg of B complex + a well rounded multivitamin.
Thanks for your reply, Dr. Lonsdale. I will try the lipothiamine from Cardiovascular Research. Is there a max dose at which to stop? I see it can get pricey with this formulation.
I have been advised by neurologist not to supplement with B6 or P5P as I previously supplemented and had B6 toxicity. Is there a B complex without B6/P5P that you recommend? I would also appreciate a multi-vitamin recommendation (also without B6).
In addition to the neuropathy and muskoloskeletal issues from the fluoroquinolone, I have severe insomnia and anxiety/OCD, and am looking to treat those as well. I have failed several prescription medications for this. I thought to try niacinamide, but am not sure. Thank you for your time and any further response.
B6 “toxicity” comes from an old paper years ago trying to wreck its therapeutic value. The maximum dose of thiamin is thousands of times the physiological dose. B complex is not toxic. Your insomnia and anxiety is the hallmark of brain energy deficiency and should respond to thiamin.
Thanks, Dr. Lonsdale. I just got the Lipothiamine.
Is there a benefit to taking benfothiamine along with Lipothiamine (perhaps continue the benfothiamine for peripheral tissue and Lipothiamine for the brain)?
I see that you have two Thiamine books on Amazon. Which is best for the layman, and is there a source for your books that are cheaper than the prices on Amazon which are quite expensive.
Thanks again for your time.
Try Lipothiamine from Ecological Formulas. Start at 100 mg/day and increase according to whether or not you experience benefit. There is no maximum dose and there is no toxicity. If symptoms get worse temporarily, stick with it. Find the article on HM that deals with “paradox”. Your problem is mitochondrial and might be genetic in origin.
Thanks, Dr. Lonsdale.
I took 100mg of the Lipothiamine today with food.
I did see the two Paradox articles you wrote and will be mindful of this.
I decided to continue with the benfotiamine as well but stepped down to 300mg from 600mg.
I did want to mention that while the fluoroquinolone resulted in immediate neuropathy and musckuloskeletal issues – now for years, I first began to notice odd health problems after taking an acid blocker over ten years ago. I think this dovetails with some of your writings. I also have been extremely sensitive to noise, getting years-long tinnitus ear damage from a single loud event even while others were unaffected. And I had a strange reaction to a short course of prednisone resulting in body-wide stiffness. This preceded the fluoroquinolone as well. In fact, I have always been unusually sensitive to both the external environment and pharmaceutical drugs.
Conflicting with my theory, though, is that I have had the severe anxiety/ocd issues for most of my life, certainly preceding the fluoroquinolone or the acid-blocker exposure.
I will be interested to see if Lipothiamine can help my diverse and systemic problems.
Thanks for your time.
Hello Dr. Lonsdale!
I would like to know if you have any idea if taking steroids could impact or deplete thiamine in the body?
Thank you,
Cathy
Probably! Steroids stimulate energy consumption.
Thank you! I was on steroids (supposedly to support my adrenals) for 9 years and it was finally so bad I was suicidal and could not function. I think the drug pushed me over the edge from lifetime, sporadic (but unrecognized) B vitamin issues to full out damage. I changed doctors, not knowing what the heck was wrong with me, unable to walk, with many, many other symptoms/side effects. She discovered I was terribly overdosed on steroids and it took me a year and a ton of patience on her part to slowly, carefully wean off of them. Thinking I was better and didn’t need them anymore I stopped the vitamin IVs and supplements and now, 2 years later I am getting sick again. I saw people discussing the SLC19A1 gene and I got out an old 23&Me report. I can’t remember the verbiage anymore but I do have an issue. I think I saw in one of these posts that you told someone when they have gene issues they need to ALWAYS supplement (I also have an MTHFR issue). I am not as sick as I was on steroids but I am sick enough not to have a life so I have to fight my way back to something resembling “normal”.
I am going to start the Allithiamine at the 50mg dose and tough it out until I can go up on it but can I do the Myer’s IVs too or should I wait until I can up the Allithiamine? I am anxious to get moving, now that I think I have a grasp of what’s been going on.
And, oh my gosh!! Where would we all be without you? I can’t even imagine how we would ever be able to figure out what the heck is wrong with us without all of this info you provide! Even the doctor who helped me get off of steroids, as wonderful as she is, has a hard time believing I have thiamine issues and that is what my symptoms are all about. I am so glad I can handle this myself. She is happy to do the Myer’s though!
Cathy
Thank you so much for this informative article! What companies produce thiamine tetrahydrofurfuryl disulfide? Is it available over the counter? If so what online retailer sells it?
I have only been able to find the one supplement by the company you referenced for allithiamine and a handful for benfotiamine. I would of course prefer TTFD for many reasons as you stated.
Allithiamine (AL) and Lipothiamine (LI) are both TTFD. The only differences are: AL is powdered so the dose can be split:LI is an enteric coated tablet of 50 mg TTFD to pass it through stomach acid to get to the small intestine where thiamine is absorbed. Both are available from Ecological Formulas on line.
Sold as Lipothiamine by Ecological Formulas in 50 mg tablets
Hi Dr. Lonsdale,
I understand that you recommend above all Lipothiamine 50mg (TTFD) manufactured by Ecological Formulas, because it is enteric-coated. However, I see that Allithiamine 50mg (TTFD) capsules also manufactured by Ecological Formulas, are much less expensive.
In this context, can you clarify whether the Allithiamine product is an effective option for supplementation for our issues as compared to the Lipothiamine (TTFD)?
Thanks for your insight.
I would like to share my Thiamine experience: as a young woman 28 years ago, I consulted a cardiologist because of stenocardia, having had endocarditis 2 years before. I also had difficulties concentrating, always had brain fog and was not able to learn s.th. new, always had some open skin on my hands and lips, my face was red and burning, and I was no more able to practice contraception via temperature, because my curve was unphysiologically chaotic, the temperature always being close to 37.0 without a cyclic pattern. I gave up to care, but did not get pregnant. I had “irritable bowl”. I felt miserable and certainly was complaining about it; the cardiologist only gave me the address of a psychologist. I was angry and resignated, but went there out of curiosity. I actually liked to talk to her, but after only a few meetings she asked me to find myself a proper doctor. I found an endocrinologist who drew incredibly much blood, confirming sarcolemma-antibodies and thiamine deficiency! Being a neurologist myself, I could hardly believe it; never had we taken thiamin-levels for comparable complaints. Well, I started eating benfotiamine, and within one week my skin was without sores, my face smooth, my mind clear, stenocardia gone and my cyclic temperature changes became physiological again. After 3 month, I was pregnant. A few years later, I drew blood for a thiaminelevel from a severely suicidal psychiatric patient who complained about being unable to perform in her job as a simultaneous translator because of brain fog and forgetfulness; she had the same skin lesions as I knew them. Her thiamine level was low, and she profited from substitution in the same way as I had. I hardly ever had a more thankful patient!
I ate Vitamin B1 for many years until I decided that I can do without because my irritable bowle was gone since I stopped eating whole grain stuff. Another few years, and I developed palpitations and started feeling somewhat depressed and resigned. I remembered thiamine and gave it a go – again helping me immediately. The palpitations were gone, the mood good, and in addition, an annoying achillodynia vanished. I have since been distributing thiamine among family and friends, and they confirm better mood and one colleague could – like myself – control her palpitations with it.
Dear Dr Ridder. I have been trying to get physicians aware of the expected results of your own treatment. I think that you would get a big bang out of our book “Thiamine Deficiency Disease, Dysautonomia and High Calorie Malnutrition” available at Amazon Books. It was written primarily for physicians,but many patients are recognizing the cause of their symptoms that have defeated their own physicians, sometimes for years.
Hello Maria!
What a great story! Can you tell us please what dosage you used to achieve this incredible result? (in mgs please?)
Thank you,
Cathy
Dr. Lonsdale, to add to my history, I am 37 yrs old. I have been recently diagnosed with hypothalamus hypofunction function due to high inflammation and leptin blamed by mold suceptible genetics. The doctor that I’m seeing is referring to the Shoemaker protocol for mold exposure. But to complicate my situation I had been given 5 weeks of fluoroquinolones and steroids at the same time of supposed mold sickness that started 10 years ago. I had a severe reaction to the fluoroquinolones as described by other “floxies”. I have major anxiety, low blood pressure, blurry vision, severe fatigue and muscle wasting on going for 10 years. I was perfectly healthy before 10 years ago other than chronic sinus problems. I suspect induced this one deficiency from the fluoroquinolones, although I can’t prove it. I need testing done.
Sorry———–I have no idea!
Dr. Lonsdale, I have read several of your articles on thiamine deficiency. I clearly have the symptoms. My biggest issue is dysautonomia. It as plagued me severely for 10 years. I was given a 5 week dose of fluoroquinolones 10 years ago and have extensive problems with my connective tissue that will not repair. My sleep is ruined. My quality of life is ruined. I have severe fatigue, high inflammation and immune system disfunction via lab tests. I’ve taken methylated b vitamins for a couple years and am intolerant. I clearly need my tissue thiamine levels checked and could use help monitoring my situation since I’m not functioning well. Which labs are currently doing the proper nutrients tests that I can have my primary doctor run to get started? Also, is there anyone in Utah or surrounding area that is competent with this issue to help me do IVs and supplements to get my health back? Supplements can only get you so far when you are as far down the rabbit hole as I am. Your help is greatly appreciated. I’m willing to drive out of state for the proper help and/ or do phone consultation with labs. If you can’t make a recommendation for someone to help me, does your latest book have all the information I need to help myself?
I would certainly suggest that you get our book “Thiamine Deficiency Diseases, Dysautonomia and High Calorie Malnutrition” from Amazon books.
Is there anyway to get this book cheaper as im unemployed
Depends on seller!!!!
I am in the 3rd week of taking allithiamine. I decided to repeat my blood tests. I always had low count of neutrophils (either neutropenia or close to neutropenia). I am fighting endometriosis and during childhood I had asthma – so I assume my neutrophiles issues may be from there.
Now, my neutrophils has increased by 10 % almost. Not sure if it’s due to allithiamine as I took other supps during these 6 months but I assume it’s because of it , as my energy level increased a lot.
Does thiamine also increase neutrophils?
Thanks a lot!!
Hello Dear dr.Lonsdale.
I just wanted to mention a point that I think would be a great importance.
This thiamine paradox for some people may be simply vitamin b6 toxicity because of the b complex that they’re are taking along with thiamin. Obviously there are many people who may reach toxic levels of b6 very easily and unfortunately there’s little info on this issue in the current medical world.
Vitamin B6 neuropathy was reported many years ago and was particularly aimed at vitamin use in general. I have used huge doses of B complex over the years and have never seen any toxicity.
Hi, Dr. Lonsdale, I need to ask something:
Can I take sulbutiamine instead of allithiamine? Both are thiamine disulfide if I’m not wrong. Allithiamine causes me intestinal pain (I have many intolerances and gastritis frequently, maybe its for garlic, i dont know), however, sulbutiamine does not hurt me. What is the difference between the two? Have both same effective? If your answer is affirmative: do I have to take the same dose of sulbutiamine as allithiamine? That is to say 50 mg sulbutiamina rising little by little up to 200-300mg?
In summary, my question is whether I can follow his guidelines and Dr. Lonsdale with sulbutiamine instead of allithiamine, or would have to change something
(I am taking with breakfast, with food. My stomach is very sensitive and I usually have this type of problem. I would be very grateful if you could resolve the doubt when you know it. It is very important for me to be able to do the treatment correctly)
Thank you very much
Yes, I have never used it but it is also an open ring form of thiamine
This could be the missing link! My 11 year old son was just diagnosed with EOE today. He’s had pain after eating for a year and a half. Hes been gluten and dairy free, prior to and was eating a standard American diet (sugar). He constantly complains of “breathing issues” where he has to tell his body to breath. He has urinary frequency since almost 2 years ago, when ocd and ADD symptoms came out of the blue.
He has the mthfr mutation and is on a ton of great supplements but I’m wondering if it’s the best thiamin. What’s your suggestion to start him on Lipothiamine? Hes 78 lbs.
Thanks for all of your wonderful info!
Please see the post on this website “Is eosinophilic esophagitis a sugar sensitive disease?”
Dr Derrick, I wanted to supplement with Thiamine but I heard it contains Sulfur. I do react to Sulfur based foods and taking Magnesium Sulfate gives me an anxiety attack. I have gut dysbiosis and issues like Anxiety, Panic attacks, Chronic fatigue etc. I am currently taking B12, D3, B6 and Nicinamine. Is there a N1 supplement that I can take which does not contain Sulfur/Sulfate? Thanks
Would Thiamin
—(as cocarboxylase [Coenzymated]) as in a coenzymated b complex be as effective as Allithiamine or Benfotiamine? I ask because I took it before but it did not seem to help. Does the “coenzymated” part mean it wouldn’t require transporters to get into cells? Source Naturals sells coenzymated B Complex.
Cocarboxylase is the active form of thiamin
Would the cocarboxylase be as effective as the Allithiamine at getting into cells?
No
Just clarifying that Cocarboxylase is available in a supplement Source Naturals sells as a Coenzymated B Complex. So would it be as effective at getting into the cells as Allithiamine?
No
Hello Dr. Lonsdale,
I understand that simple sugars are contraindicated for thiamine deficiency unless from whole foods. I wonder about the supplement D-Ribose, though, since it is supposed to assist with mitochondrial energy. Could you also comment on the use of sodium or potassium bicarbonate topically (in baths) or oral supplementation in regards to thiamine deficiency. Thank you!
Terminology is important in understanding my answer. For example, what is meant by “whole foods”? It should be replaced by the term “natural foods”. But the distortions in linguistics so often confuse the issue. The food industry is using the term “all natural” that is completely meaningless within the context of its use. I see no reason for supplementing with d-ribose, sodium or potassium bicarbonate.It is very clear to me that our advance in medicine has to be by a comprehensive understanding of cellular biochemistry. It has long been said that “a little knowledge is a dangerous thing”. It will take a very long time before the medical profession picks up the slack. Although we know a lot, it is overwhelmed by our ignorance and our progress demands sophisticated research. I read new articles in the medical literature that provide highly sophisticated nutrient based medicine every day and most of them are coming from countries other than America. Let nobody read this blog with the idea that all the answers are available. The complexity of a single cell is simply mind-boggling. In my clinical experience I have tried to keep it simple and I became aware that thiamine used as a drug rather than simple deficiency replacement was a huge advance in itself. For those reading this blog, I sincerely recommend obtaining the book by myself and Dr. Marrs. “Thiamine Deficiency Diseases, Dysautonomia and High Calorie Malnutrition”. Much of it is readable by the intelligent public and answers some of the questions that keep cropping up here. The highly technical areas can be skipped or studied, according to the experience and knowledge of the reader.
Dr. Londsdale,
What can be the negative effects of taking Allithiamine with an SSRI?
I can’t stop the SSRI right now and need the Thiamine, what am I to I do?
Would Lipothimaine be better as I’d prefer this due to it coming in tablet form so I can cut it down and start small. The Allithiamine only comes in capsules.
I see you and poster “Jason” mentioned Refeeding Syndrome. This is what I am trying to treat per se with the Thiamine. I posted those articles in my post below that state that Thiamine deficiency during refeeding a malnourished patient could cause similar symptoms to Refeeding Syndrome. Are you stating that taking Thiamine for a severe Thiamine deficiency causes Refeeding Syndrome?? In these articles it states Thiamine supplementation resolved their electrolyte loses and other clinical Refeeding Syndrome symptoms.
Yes to Lipothiamine
Can you explain what are the dangers of taking an SSRI and Thiamine concurrently?
Also would a Thiamine pyrophosphate/TPP supplement be as effective as Lipothiamine?
Thank you.
No
Vitamins, including thiamin, are known as “prodrugs” because they enhance the activity of a drug
Dr Lonsdale, when you say remove sugar in all its forms could you specify? Does that mean fruit as well? Does it mean all starches even vegetables? I looked up foods with high b1 and have been eating more red lentils, oatmeal, etc, but these foods are high in carbohydrates.
I mean the products of the food industry. Of course sugar is found in fruits and vegetables and that is the way that we should be taking it. We have become extraordinarily artificial in life and the further we get from our own biology the more dangerous it becomes. It is probably the fiber in the fruits and vegetables that modifies the way the sugar is metabolized. That is why the food industry has produced products that contain fiber and they do not work. Civilization is virtually our enemy. Any food that God has made is okay. Man is an omnivore, meaning that he can eat meat as well as vegetables and fruit. All you have to do is to look at our teeth. We have “cutters” to cut food, in company with pointed teeth, designed for tearing chunks out of a carcase and “grinders” to grind down plant products. This is how our ancestors were equipped as they emerged as Homo sapiens. Mcdonalds and Coca-Cola were in the future and represent the modifications that appeal to our hedonistic side.
I was eating a lot of watermelon over the summer because it was in season and low in oxalate. I started noticing that I would get burning sensations in my feet and legs after eating it so I stopped.
@Chandler Marrs is there a template for checking that Snp?
Charlotte are you referring the thiamine transporters or other nutrient transporters? If so, they are called solute carriers, SLC for short, there are almost 400 in total. The ones involved with thiamine and other B vitamins are the SLC19A2 and SLC19A3, folate uses the SLC19A1.
Hi may I ask if any of the thiamine transporters in the gut are copper dependent? Thank you
No
Dr. Lonsdale, were you able to read my post from August 3rd?
Thank you for your response to my original post.
Another question though: A
re there any contraindications to taking Allithiamine if one is also taking an SSRI like Lexapro?
yes
Chronic B1 and B3 deficiency
Hello,
I have chronic functional B1 and B3 deficiency per SpectraCell Micronutrient Panel. I have tried multi and B-complex, but these didn’t help improve B1 and B3 per follow-up SpectraCell testing. I have started reading many of your articles about B1 and have started taking Lipothiamine 10 days ago to cover B1 along with multi w/ B’s, B-complex, electrolytes and magnesium.
Any special considerations for B3 deficiency? Is Multi w/ B’s and B-complex enough? Is there a special B3 form I need to take to overcome possible genetic or transport issues?
My current oral B3 dosage is 150 mg per day via 35 mg Niacin and 115 mg niacinamide.
Thanks in advance.
You are doing alright. It would be helpful to others on this website to describe your symptoms and why you had the tests done in the first place
It’s been a long 3 year journey, but my symptoms are best summarized as POST/CFS.
In early 2014 I started experiencing fatigue, aches, and sinus issues. I assumed allergies, infection or cold, but it never went away. Just waxed and waned. I bounced around doctors trying to figure it out, but no doctor could find anything and many just wanted to manage symptoms. About 6 months into this I started having anxiety, depression and panic attacks which I had never experienced ever in my life.
Eventually, doctors started saying it was all in my head and I need to see a psychiatrist. I stumbled into a Functional Medicine psychiatrist office and she was big on nutrition and other less than main steam illnesses. She ran more tests than all of my other doctors combined. Through this testing we suspected a nutrient deficiency component, but struggled to nail it down. One and half years later I stumbled across a very experienced nutritionist and he recommended SpectraCell Micronutrient testing. As a result, we now know that I have low levels of B1, B3, and borderline B6 and B9 deficiencies. Also, a methylation pathways panel has confirmed low cysteine, taurine, and glutathione.
We are trying to fix these deficiencies, but we are struggling. I started Lipothiamine 2 weeks ago, because Thiamine HCl and sublingual cocarboxylase didn’t help my B1 deficiency. It’s been a difficult start. Increased energy, slight POTS and IBS reduction; however, restlessness and anxiety has increased significantly. Also, increased pins and needles, and poor sleep. Maybe this is the paradox that is mentioned in several articles. This paradox is incredibly unpleasant and I understand why people stop treatment.
I wish there were more stories on the paradox. I feel like I am flying blind.
You ARE “flying blind” because very few physicians know about “paradox”. It also has been mentioned on this website as “refeeding syndrome”. When you have been deficient in thiamin, in particular, for a long time, the paradox is apt to be prolonged because you are resuscitating a mechanism that has been broken. It is too difficult at a technical level to explain refeeding syndrome or paradox. It is to do with an explosion in oxygen utilization. The longer you have been deficient, the longer the paradox and it is entirely and completely unpredictable. My experience over many years is that it is the best prediction of ultimate success. It is encoded in the saying “there is no gain without pain”. Jason has evidence of multiple B complex vitamin deficiencies and he would benefit best from intravenous vitamins or multiple injections of B complex.
Thanks Dr. Lonsdale for your reply. Would it be possible to post one or two new articles on the “paradox” and “refeeding syndrome.” One article could focus on educating us on the science behind paradox and refeeding syndrome. a second article providing deeper and more detailed examples of patient experience during paradox and refeeding syndrome.
I found two articles on refeeding syndrome which may be helpful for others:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2440847/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2654033/
Thanks in advance.
Jason, that is a great idea. Much of this is covered in the book and encourage everyone to purchase and read the book. Nevertheless, a few more posts on the topic would be useful.
I wonder, if also, you might consider pulling all of your comments together into a post about your experience. Include your symptoms, the tests, what you’ve learned, etc. These case stories are critically important to build awareness, and frankly, to build the knowledge base regarding this issue. Please let me know if you will write your story. I will help edit. Here’s the link to contact me. https://www.hormonesmatter.com/write-for-hormones-matter/
See my post from August 3rd below. I posted links to several medical articles stating Thiamine supplementation resolved symptoms of Refeeding Syndrome in patients.
Jason,
See my post from August 3rd below. I posted links to several medical articles stating Thiamine supplementation resolved symptoms of Refeeding Syndrome in patients.
Hello Dr. Lonsdale,
What are your thoughts on anticholinergic drugs in relation to thiamine deficiency? I currently take two, sertraline and doxylamine succinate. It appears that anticholinergic drugs and thiamine affect acetylcholine levels.
I don’t know anything about the drugs because I never used them. Thiamin deficiency will result in loss of cholinergic neurotransmitter drive. I cannot think of anything less likely to help you by taking an “anti-cholinergic drug” because cholinergic drive is extremely important in many aspects of brain metabolism
Is there any reason why you don’t see IM injections of TTFD forms of thiamine? Is it because they are so effectively absorbed orally or because they aren’t water-soluble enough to efficiently take as an injection?
TTFD is not approved by the FDA. I had an independent investigator license from 1973 and treated hundreds (if not thousands) of patients. Why can it be used to treat so many different conditions? Because the famous researcher, Hans Selye showed that human diseases were all “diseases of adaptation” and that they were ALL due to lack of energy. TTFD stimulates the cellular machinery that produces energy and might even work in some genetically caused diseases because the relatively new science of epigenetics has shown that nutrition can influence genes.
Dr. Lonsdale:
I am experiencing an electrolyte imbalance (low phosphorus and potassium) after a period of fasting during Lent. I began eating normally again and experienced what I originally thought was Refeeding Syndrome. I had been diagnosed with this in the past and like this time, despite treatment and supplementing with up to 3,000 mg of Phosphorus and 160 mEq of Potassium, my blood levels are still low.
I have weak, heavy muscles, edema and can’t walk more than 50 feet without resting or else my body feels like heavy jelly. Last time I had this issue in 2015 I tested very high for urinary loss of Phosphorus and Potassium. My Nephrologist thought I had kidney damage from a toxin but he also thought it could be the massive amounts of supplements I was taking for the Refeeding Syndrome. This time around my urine was not tested.
I researched online and believe God led me to several medical articles stating that Thiamine deficiency during the refeeding phase could cause renal tubular dysfunction resulting in electrolyte losses via the kidneys. So electrolyte values during the refeeding phase after fasting could be Refeeding Syndrome’s intracellular losses -OR- urinary losses from Thiamine Deficiency.
I decided to purchase B1 tablets in the form of Thiamine Hydrochloride and started 8 days of 100 mg. I also took Mg 400mg and a multivitamin. On day 6 I experienced increased energy as well as complete resolution of (8 weeks of!) extreme muscle weakness and heaviness, shortness of breath on exertion, absolutely no energy to walk more than 50 feet, as well as edema. I had energy into late into the night.
That only lasted for 2 days and then I experienced extreme tiredness and symptoms returned, albeit not as bad as before. Once I stopped the B1 the extreme tiredness left. I tried it again and the tiredness returned. I know that B vitamins can cause unusual tiredness in some people.
I really believe I am onto something here though. I have decided to try an active form of B1 and am not sure if I should go with Alltiamine or Coenzymated B-1? Do you have any suggestions?
The patients in the articles were repleted after a short period of days to weeks and had resolution of their electrolyte imbalances. I am concerned though because I have been in this condition for over 2 months now and if it is in fact Thiamine deficiency I worry that maybe there is permanent damage to my kidneys.
The medical articles are pasted below if anyone is interested in reading about this.
“Thiamin and folic acid deficiency accompanied by resistant electrolyte imbalance in the re-feeding syndrome in an elderly patient”
http://apjcn.nhri.org.tw/server/APJCN/26/2/379.pdf
“Thiamine Therapy and Refeeding Syndrome in Extremely Low Birth Weight Infants”
http://www.casereports.in/articles/6/3/Thiamine-Therapy-and-Refeeding-Syndrome-in-Extremely-Low-Birth-Weight-Infants.html
“Acute thiamine deficiency and refeeding syndrome: Similar findings but different pathogenesis”
https://www.ncbi.nlm.nih.gov/pubmed/24985016
Low potassium occurs in thiamin deficiency. I would advise you to keep up with Allithiamin, magnesium and a multivitamin on an indefinite basis
Could any of the forms of thiamine be applied topically with either water of fat mixed with it if one could not tolerate the oral forms i.e.: due to digestive issues like chronic malabsorption and diarrhea.
Are there any companies that make thiamine as a cream or transdermal gel? Everytime I take b-vitamins orally, I get serious diarrhea that last very long, and eventually it makes my b1 deficiency, along with other b deficiencies, worse.
Ecological Formulas in CA makes Authia. Getr it via your physician
Ecological Formulas make Authia cream. Get it via your physician
I’ve been having problems waking up with a dry mouth for years, and recently air hunger, I thought was due to apena, however ENT could not diagnose me with sleep apnea after reading sleep study. He could not figure out way I was suffocating at night as he never had a skinny patient before. I found your article about pusedo-hypoxia after I talked to the ENT.
This spring I was diagnosed with a B1 genetic deficiency homozygous SLC19. I ordered B1 Benfotiamine, HCI, and sulbtiamine and have been experimenting in dosages up to 500 mg with regular dosing of the other multi- B’s. I just ordered the TPP form. The 500 mg B1 dose was recommended by a hypothyroid clinician, who has seen marvelous energy improvements. At this large dose my suffocating went away immediately on the first night, and now I wake up only 2 times a night instead of 6 times. I have been on this protocol for only 7 days and surprised with the fast results.
I have Hashimoto’s and have not felt well under all the different meds I’ve tried over the past 5 years. I believe my condition is due to B1 deficiency, and the symptoms are getting more severe now that I’m 52.
I would like to try IV injections until my breathing problem is cured. There isn’t a lot of information on B1 IVs. Is there a therapeutic dose or IV drip to start with muscle shots as maintenance to continue? Or would you just recommend I use oral B1? I eventually develop tolerance to oral supplements and they stop working all together.
Thank you for sharing your work with us all.
This is a marvelous example of thiamine deficiency. Notice that the symptoms were confused with sleep apnea and probably with Hashimoto. If you read and understand the post above, you will come to the conclusion that the best supplement is the one that has the chemical name of thiamine tetrahydrofurfuryl disulfide (TTFD) and sold in the United States as Lipothiamine available from Ecological Formulas. Notice also that Karen got immediate relief for some of her symptoms but not all.She expected her “breathing problem” to be relieved. All she needs to do is continue with the large doses of oral thiamine derivatives because they do not need the transporter protein that she tells us is missing. She should also add large doses of magnesium because that works with thiamine. A well-rounded multivitamin is also a good idea because thiamine does not work on its own. It is a team member. I must emphasize here that the compromise of automatic breathing is due to thiamine deficiency in the lower part of the brain. The dry mouth in the morning is almost certainly because thiamine deficiency stimulates the sympathetic component of the autonomic nervous system so I would expect some components of the fight or flight reflex.Treatment of long-term thiamine deficiency is quite different from the use of a pharmaceutical drug. It is relatively slow as the system reconstitute itself and patience is required
I read you comments on increasing magnesium potassium aspartate and magnesium salt.
Could you comment on foods and supplements that destroys B1 transport? My lab cited blueberries and raw fish as antithiminase. I was eating berries everyday because of low sugar content and taking resveratrol and 88% dark chocolate, which happen to be the only two of the few foods that I like that I’m not allergic to. I do not eat sushi anymore, because of toxins and white rice. I have an intolerance or sensitivity to many common foods. I’m paleo 80% of the time and do not eat dairy or gluten. Whenever I eat too much of one food, I develop a IGE or IGG response, I’m sure this is due to the B1 deficiency and hoping the inflammation will go down in time. I don’t have enzyme activity to breakdown sulfur, SOD and histamine nor transport folate, so vegetable nutrients aren’t being absorbed. I read
Vitamin C foods help with absorbstion, but I’m allergic to citrus fruits and nightshades. Would something like Perque Vitamin C powder compensate for times I ingest polyphenols?
I try to stay at 20-25 grams of sugar a day, I have insulin resistance. Is this still too high?
I was able to wipe out Candida Krusei but I do still have yeast overgrowth (white toungue) due to IBS-C. I had elevated oxalates but not sure if I still have it. Would taking calcium citrate help breakdown oxalate foods? It’s been extremely difficult eradicating my yeast infections.
An interesting topic would be the quality of lab testing available. I have taken Genova OAT and Great Plains labs in the past, I took the Spectrecell lab most recently which was the one that measured my B1 and aspartate deficiency and low glutathione, big markers in my case.
I will be taking your articles to my Orthomolecular doctor that is out of state in a few months, and he will be extremely pleased at the information available here. My GP is excited to monitor this new way of treating my thyroid problem.
Point number 1: there is obvious confusion here. Thiaminase is an enzyme that occurs in certain bacteria that live in the human intestine. It has the capacity to break down dietary thiamine. It has nothing to do with transporters. Thiaminase also occurs in the intestines of fish and in several plants. Note that when we eat sushi we do not consume the intestines of a fish. As far as I know blueberries do not contain thiaminase. Point number 2. There are several proteins known as transporters, several of which transport thiamine into our cells. They are made in the body and are under genetic control. If one or more of the thiamine transporters is missing, it is a source of body and brain cellular thiamine deficiency.Point number 3. A level teaspoonful of sugar would be 5 g. So taking 20 to 25 g would be the equal of 4 to 5 teaspoonfuls. This would easily be the cause of thiamine deficiency, particularly if there is a transporter problem.Point number 4. For those interested, there are many posts on this website that discuss various aspects of thiamine metabolism and I do not need to repeat them here.
Karen,
I wanted to share some info based on your second post, where you mentioned blueberries. In 1999, The World Health Organization published a report on thiamine, which indicates that polyphenols in blueberries are thiamine-antagonists (or anti-thiamine factors), which is different from a thiaminase found in raw fish. these thiamine-antagonists are found in other foods, such as red cabbage, tea, red beets and others. Quercitin is another thiamine-antagonist that is used that is used as a supplement. It was recommended to me years ago for allergies, and I know recognize that some of the symptoms I experienced while taking it were likely due to thiamine deficiency.
I would be interested to know if large doses of the polyphenol resveratrol is an anti-thiamine antagonist. I don’t believe it was used as such when the WHO published that report in 1999. Perhaps, Dr. Lonsdale could answer that.
Not sure if I can share the WHO link to thiamine here. I’ll try: http://www.who.int/nutrition/publications/en/thiamine_in_emergencies_eng.pdf
Let me make it as clear as possible. Natural, edible foods provide the correct fuel to the human body. Resveratrol is a substance contained in some foods and is part of the genius of that food. If you remove an active principle from a herb it has an entirely different action from that of the whole herb. This is the frightening mistake of the food industry. Of course, we know only too well that not everything created by Mother Nature is good for us. Some berries and mushrooms are frankly poisonous and we had to learn that the hard way as a species. I keep seeing posts, saying that taking B1 “upsets me”. This has given rise to a post called the “refeeding syndrome”. If you have been deficient in vitamin B1 for a lengthy period, you have to be very cautious in replacing it. It requires very small doses to begin with and you build up as the adverse symptoms gradually cease. Unfortunately, because of the ignorance in the medical profession concerning the basics of normal nutrition, it is difficult to find a physician who understands the problem and how to correct it.
I am struggling to understand all the information about thiamine and how to supplement it etc so please forgive me if I’m asking what seems obvious. I am trying very hard to educate myself but I don’t seem able to retain information very well at the moment. I believe I am deficient in thiamine (POTS, CFS, more recently apathy, nausea and lack of appetite and lack of cognition that had me fear that I was losing my mind). I struggle to supplement orally because I get increasing gut pain from this. Is this what you mean by people saying B1 “upsets me”? You mention a post called “refeeding syndrome” are you referring to comments by site users or a post written by a site contributor? I tried to search for it but came up empty.
At the moment I can only manage oral supplementation every 5 or more days without getting worsening stomach pains. This has taken the edge off the worst of my symptoms but is not really enough to replete me I suspect. I don’t know how to proceed. I am in the UK and cant source all forms of B1 that have been mentioned. I have Benfothiamine and the HCl form. When you mention starting with very small doses what sort of doses do you mean? Also what are the typical adverse symptoms? Would my gut pain be one such symptom? I do get similar gut pain from other supplements and many foods so it is not only B1 that causes it.
Are there key posts that would be most helpful for me to read? A lot of questions I know.
My main difficulty is how to get the B1 inside me in a consistent way. Any help or guidance you can give would be most appreciated.
I need to know your age and a history of your illness. I also need to know your intake of sugar and whether alcohol is an important item.
I don’t drink alcohol at all. I was low carb for several years but in an effort to put on weight and stimulate my metabolism I started increasing carbs. Mostly potatoes since I cant eat grains at the moment and sugar. For a few weeks I was probably eating 60 – 80g sugar per day (previously none except occasional honey).
In fact it was upping the carbs that caused the nausea, loss of appetite, apathy and cognition problems. I stumbled across an article about B1 and it seemed to fit.
Once I realised what was going on I cut the sugar down to 10 – 20g per day. I haven’t cut it out completely at the moment since I struggle to eat enough on a pretty limited diet.
I’m 48 and have had chronic fatigue since 2004. Major worsening 2008 – virtually bed ridden. Slowly gained some strength back but still pretty severe.
Prior to 2004 had anorexia (including binging and purging for 10 years or so) and severe depression.
Diagnosed with POTS 2012 though largely resolved now.
Have had low blood pressure since at least mid 20’s Typically around 90 / 60 though has been lower.
Many thanks
I don’t know if it’s of any relevance but I was diagnosed with Hereditary Haemochromatosis 2 years ago. I am now in maintenance so venesections are less frequent. I’ve had 12 or so units of blood removed over the last 2 years.
I would be interested in your story
Sugar is your undoing! I would expect that you have extremely severe thiamin deficiency because you have some of the symptoms of beriberi. You have to give up sugar absolutely completely in all its forms and you will need large doses of thiamin and magnesium and a multivitamin to recover
Thank you so much for replying.
I must admit I suspected that adding in the carbs was revealing a B1 deficiency that I previously wasn’t aware off. When you say large doses, what sort of figures are we talking about? This is my problem because I get stomach pain when I take oral B1 several days in a row (it gets progressively worse each day) and I am worried that if it is inflaming an already highly sensitive gut will I even absorb the B1? Is there another way of getting it in me? I have a similar problem with magnesium. At the moment I have epsom salt baths most days in an effort to get magnesium in me.
Could I dissolve the B1 in water or DMSO and rub it on my skin?
I feel stuck in a catch 22, I know I need the B1, but taking it makes my gut worse.
Dr Lonsdale
I have recently had the transkelotase test done at Biolab
http://www.biolab.co.uk/index.php/cmsid__biolab_test/Vitamin_B1_(Thiamine)_-_transketolase_activity
My test result is 1.09 which is the ratio of activated to basal activity. Anything below 1.15 is considered normal. I am now totally confused because everything I have been experiencing leads me to believe I have a thiamine deficiency and this test seems to be saying otherwise.
When I first came across all this info on thiamine I started supplementing and the worst of my mental confusion, nausea, loss of appetite went away within a few days. I stopped the B1 because of gut pain and the symptoms increased again, I then restarted the B1 and they decreased again. (Unfortunately I had been supplementing for a bit before performing the test but intermittently) I am now using the Authia cream since I struggle with oral supplementation. I have noticed that I feel really tired after applying the cream for most of the day. I seem to remember reading that this would also suggest deficiency as otherwise supplementing would have no effect.
I’m inclined to believe my response to thiamine rather that the test result but I was wondering if there is any explanation that would explain the result.
I had a RBC mg test run at the same time and that showed a slight mg deficiency (1.92 with a ref range of 2.08 – 3.00 mmol/l). Again I struggle to supplement with mg as I can’t take it orally. I rely on epsom salt baths and in the past mg oil on my skin.
I would be very grateful for any insight or advice you have to offer.
Many thanks
Of course I can’t find it now, but there was a case report of an anorexic patient who needed both thiamine AND sugar in order to gain weight. It was on the sciencedirect.com website where many of your papers and chapters of your book can be found.
Is it possible that when one is so deficient in ‘everything’, that a little sugar is the easiest to digest, and helps rebuild glycogen levels?
Thanks in advance.
Nicky since you are in the UK – you have access to Metabolics.com which make Thiamine Pyrophosphate TPP which is a very bioavailable form of Thiamine. You can buy the liquid form or capsules. The product is the cleanest you can find and I buy it all the way from the UK – I’m from Australia. You might want to ask Dr Lonsdale about dosing. I would also love to know. This type of Thiamine seems to suit my 10year old son as he does not tolerate any sulfur supplements. I do have an order in for Authia cream which i’m hoping will help him. My son I believe is extremely Thiamine deficient and symptoms started showing as young as 12 months – no dr has ever pointed me in this direction – he had classic symptoms , chronic vomiting, stiff neck, dilated pupils and a drunken sailor walk when he was learning to walk at 14 months. He has a severe autism diagnosis. I’m hoping it is not too late to reverse the damage completely..
Hello!
What is considered a “large dose” of thiamine and magnesium please?
Thank you!
Thiamine from health food store 100 mg. Lipothiamine from Ecological Formulas 50 mg
Hi Karen,
I’m sorry to hear that you are experiencing these problems. Your post caught my attention because you mentioned that you were diagnosed with a B1 genetic deficiency homozygous SLC19. If you see my post, please explain how you got that diagnosis and what doctor you worked with to discover it. I have my raw data from 23 & Me, but I’m looking for someone to advise me on it.
I discovered that I was thiamine deficient a few years ago and it’s likely a genetic issue. I don’t respond well to the usual forms found in supplements, like thiamine mononitrate. I take Lipothiamine, magnesium and a B-complex. Currently, I’m “experimenting” with intra-muscular thiamine injections, too. IV’s tend to be a lot more expensive.
I wish you good health!
Run the 23andMe reports through livewello.com. They do a more comprehensive SNP report. It’s a little complicated by provides a wealth of information, including defects in the SLCs. It cost an additional $20.
snpedia, and promethease only have slc19a1, and 2 but not disease causing mutations in them. I will try live wello thanks.
Realised I did it a while ago. They have only one of the transporters, and likely 0 of the known pathogenic mutations in it. It is a rare disease so that is not surprising.
Karen : do you have the Snp #? Thank you
Dr. Lonsdale, your blog changed my life – and probably extended it. Thiamine deficiency plagued me for many years and supplementation with Benfotiamine was a rapid miracle cure for me. By chance, a person mentioned Sulbutiamine to me today. Apparently, it’s benefits are getting attention from doctors. You may wish to add it to this important article.
Dr. Lonsdale, your blog changed my life – and probably extended it. Thiamine deficiency plagued me for many years and supplementation with Benfotiamine was a rapid miracle cure for me. This is another important post. By chance, a person mentioned Sulbutiamine to me today. Apparently, it’s benefits are getting attention from doctors.
Sulbutiamine is a disulfide derivative of thiamine. I recommend reading the post above carefully to see why the disulfide derivatives are the best