When you have a hammer, everything becomes a nail, or so they say. I worry about this as I dig deeper into mitochondrial dysfunction. Could all of these disparate symptoms and conditions have their roots in the mitochondria? Could it be that simple? Perhaps. More and more, as I search for explanations for the devastating symptoms that so many of our readers report, the research I find points to mitochondrial dysfunction. Sure, changes in gut microbiota and function are apparent and often related and certainly immune dysregulation is a component of these illnesses, but the underlying connection among these disturbances seems inevitably and inextricably linked to dysfunctional mitochondria as the central hub of illness. Heal the mitochondria, heal the body is quickly becoming my new mantra.
Mitochondria as Danger Sensors
Some researchers argue that the mitochondria are the danger sensors for host organisms; having evolved over two billion years to identify and communicate signs of danger to the cells within which they reside. The signaling is simple and yet highly refined, involving a series of switches that control cellular energy, and thus, cellular life or death. When danger is present, energy resources are conserved and the immune system fighters are unleashed. When danger is resolved, normal functioning can resume.
If the danger is not resolved and the immune battles must rage on, the mitochondria begin the complicated process of reallocating resources until the battle is won or the decision is made to institute what can only be described as suicide – cell death. Cell death is a normal occurrence in the cell cycle of life. Cells are born and die for all manner of reasons. But when cell death occurs from mitochondrial injury, it is messy, and evokes even broader immune responses, setting a cascade in motion that is difficult to arrest.
And, if the on the battlefield, the host army is understaffed and under-resourced, no matter how hard the immune fighters battle, the fight will be lost, maybe not immediately, but eventually. All sorts of mechanisms will be employed to reallocate and reinforce needed battlements, but they will be for naught, further depleting already scarce host resources, until the decision is made, within the mitochondria, to begin pulling back, withdrawing, and ultimately casting the final orders of cell death.
It’s not Autoimmunity, but Impaired Immunity
I never much liked the war model of health and disease, but it seems to work well as metaphor for immune functioning, as it is far more illustrative and useful than the self-versus non-self-characterization. Really, what army with two billion years of experience, one that contains all of the memories and skills of battles past, would misidentify itself and begin broad scale fratricide – kill itself and its brethren for no other reason but mistaken identity and do so for years on end? Sure, there can be errors, over compensation and other weaknesses in the immune system, but not continued aggression towards itself in some maladaptive response. That makes no sense and contradicts the very notion and function of an immune system – to keep the host organism alive and well. Indeed, when we consider the trillions of microbes – clear non-self entities – that live inside and upon us, the idea that the immune system evolved simply to kill the non selves seems laughable. And so, I reject the concept of autoimmunity, not because the patients who suffer from continued immune system activation are not ill, they are, but because the concept of autoimmunity belies the very nature of immune function and severely limits possible approaches to recovery.
The Naming of Things
Many of you might be thinking ‘what the heck does what we name things have to do with understanding illness?’ Well, the language and the characterization of disease impacts therapeutic choices. In a system where autoimmunity dominates the discussion, survival is predicated on suppressing the invading immune army. Consequently, most therapeutic options for autoimmune disease are immunosuppressant, and mostly they fail. In contrast, if one characterizes immune function by its ability to protect and sustain life by fending off dangers or threats to survival, be they self or non-self, it does not matter, then we can be open to finding causes for those failed battles. We can ask questions like: what resources are missing that would allow the immune army to fend off the danger once and for all or what could heal the damaged cells, scavenge toxicants and oxidants or re-calibrate mitochondrial energy production? When we re-frame the discussion in this way, we open the door to a deeper understanding of health and disease. It is from this perspective, one that says chronic immune activation is not a disease itself but a symptom of an on-going and failing immune battle, that we can get to the mitochondria as the central hub for chronic ill-health.
Evolution and the Mighty Mitochondria
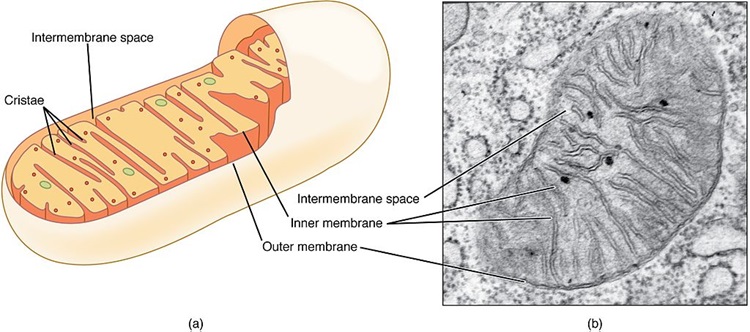
Mitochondria are interesting little buggers, having evolved from the very parasites our immune system sought to protect us against. Called symbionts, the mitochondria were microbial intruders swallowed by the host. In a brilliant move of survival, they somehow convinced the host organism not only not to kill them but to let the mitochondria, a parasitic intruder, run the host’s energy supply. The mitochondria proved their utility and developed a symbiotic relationship with the cells within which they resided. Over time, mitochondria developed a myriad of intricate communication and resource allocation mechanisms to ensure not only their survival but that of their host organism. And so, in many ways, the mitochondria evolved as part of a cooperative and collaborative ecosystem; one in which they sense and communicate danger to the rest of the organism, and if need be, initiate the final death programs; something, they should be loath to do, since their survival depends entirely on host survival.
Clearly though, and from the very beginning, the mitochondria positioned themselves as the brains of the operation. Mitochondria control energy. There is no other resource more important to the living organism than energy. Consider the most consistent sickness behaviors across all illness include, lethargy, fatigue, sleepiness, often followed by muscle and body aches, anorexia or the loss of interest in eating. The reduction in energy is purely a function of mitochondrial resources. The achy muscles are moderated by mitochondrial retractions of energy and the loss of appetite too, mitochondrial diminishments – recall the orexin/hypocretin system. By whatever pathway, declining mitochondrial energy production arises when danger signals, or more appropriately, cell damage signals are communicated. It is then that the immune armies are activated and inflammation sequences unleashed.
What Does Mitochondrial Dysfunction Look Like?
Everything and nothing at the same time. Mitochondrial dysfunction doesn’t lead to one, clear cut disease, even when there are clear genetic markers, but predisposes one to everything. Where mitochondrial damage is felt and the subsequent immune events present is complex and dependent upon the interactions among the host organism’s innate predispositions, environmental exposures and nutritional status, with the latter two significantly influencing each other. The microbial composition of the host, especially in the gut, but also on the skin and the various mucous membranes that interface with the outside world, can also moderate or trigger the danger signals that lead to mitochondrial dysfunction. More often than not though, mitochondrial damage is felt where oxygen demands are greatest, the brain, the heart, the gut, the muscles. Diseases that are currently identified discretely might all have common symptoms – the mitochondria.
Certainly, chronic fatigue should be considered mitochondrial in nature. I don’t think there is a more clear-cut example of mitochondrial dysfunction than severe fatigue, muscle pain and weakness. The question becomes, from where does the dysfunction originate and how can it be fixed or healed?
Migraine, seizures, ataxias and other neurological disorders are emerging as mitochondrial, particularly was more work is done on the hypocretin/orexin system.
Autonomic dysregulation, recognized under the umbrella as dysautonomias are mitochondrial in nature.
Thyroid dysfunction is likely mitochondrial in nature; the interaction between thyroid hormones and mitochondria is direct. Given the mitochondria’s role in steroidogenesis, other hormone systems are likely modulated by mitochondrial functioning.
Research is emerging suggesting that gastrointestinal disturbances, particularly those of dysmotility like IBS, gastroparesis, constipation and pseudo obstruction but also anorexia are mitochondrial in nature. Indeed, the GI system has its own nervous system, called the enteric nervous system. Only 10-15% of GI motility is controlled from brain’s autonomic system. The rest is controlled on site by the enteric system, mostly from cells called the interstitial of cells Cajal – the smooth muscle cells that propagate contractility and rhythm and form the gut barrier between the inside contents and the rest of the body. The mitochondria control energy usage and production here. Mess with these cells, diminish oxygen usage, pull back energy production and all sorts of things go wrong. We can get ill-timed contractions or no contractions at all, making the movement of food stuffs through the GI impossible. Poor absorption and metabolism of nutrients, and increased permeability of the tight junctions allowing for the leaky gut scenario common in many chronic conditions, become prominent and are also symptoms of mitochondrial dysfunction. Even anorexia, the will to eat, can be disturbed significantly by mitochondrial damage.
And I suspect, although I have no evidence to support this claim, in women, mitochondrial damage can express itself in the reproductive organs, especially when oxygen demands are greatest, menstruation and pregnancy. Consider the increasingly painful muscle contractions for some women involved in shedding the uterine lining during menstruation, just as diminished oxygen and energy in other muscles begins the cycle of lactate production and buildup that initiates pain, so too might this happen in the uterus. As mitochondrial deficiencies persist, uterine dysfunction would grow and compensatory immune system mechanisms increase until the compensatory mechanisms take a life of their own. When we consider the mitochondrial influence on GI motility, their influence on uterine function is not difficult to imagine. I have an inkling that endometriosis, the excessive growth of endometrial cells first within the uterus and then in regions of the body where they ought not be, is a protective mechanism, albeit an aberrant and problem causing one, that indicates increased mitogenesis and cell growth as a compensatory reaction to some original mitochondrial inadequacy. How this might happen molecularly provides some intriguing possibilities.
Immune function and inflammatory reactions are directly controlled by mitochondrial signals of danger and so to the extent we see chronic inflammatory conditions, one can look towards mitochondrial resources and the ensuing danger signals for clues towards reducing these reactions. While much of the clinical research is nascent, more and more clinicians, often from disparate specialties and sometimes without recognizing the immune-mitochondrial connections, have made great inroads towards healing and restoring mitochondrial function through diet and nutritional supplementation paired with the reduction and removal of environmental and medical toxins and dietary inflammasomes.
Is Everything a Nail, When One has a Hammer?
Maybe, but I can’t help but thinking that this mitochondrial hammer might be the one to hit the nail on the head and finally make some inroads towards reducing the suffering and burden of chronic disease. Only time will tell. For the moment, however, this is a hammer that deserves more recognition and whether it turns out to be the final clue or not one thing is clear, mitochondrial health is critical for human health. Deny the mitochondria their due and chronic, complicated illness will persist.
We Need Your Help
More people than ever are reading Hormones Matter, a testament to the need for independent voices in health and medicine. We are not funded and accept limited advertising. Unlike many health sites, we don’t force you to purchase a subscription. We believe health information should be open to all. If you read Hormones Matter, like it, please help support it. Contribute now.
Yes, I would like to support Hormones Matter.
Image credit: OpenStax, CC BY 4.0, via Wikimedia Commons
This article was published previously on Hormones Matter in June 2014.










Thank you for republishing this article. I wonder if you consider Ehlers Danlos Syndrome to come under the mitochondrial dysfunction umbrella? I am having some particularly difficult health challenges at the moment as I have EDS and dysautonomia, and have been suffering particularly badly since my second C vaccine last August. I had been taking thiamine for a number of years and stopped as, until getting the vaccine, my symptoms were quite controlled. Now all hell has broken loose in my body and having now read your excellent book, I started taking thiamine again, but this time I’m getting paradox symptoms that I didn’t get before. I assume, with information gleaned reading through various posts, this is because I’m more deficient than ever?
I consider all disease processes to involve the mitochondria. The stress of the vaccine likely threw you back into deficiency. The paradox symptoms could indicate that need more thiamine and/or that you missing other cofactors. Unfortunately, it will require some trial and error to figure out. In either case though, it will likely be miserable until you get to the other side.
Thank you for your reply. I am taking 100mg thiamine, B complex, and magnesium and gradually beginning to feel the benefits again. I still have a long way to go though.
I’ve been following one of the top trainers for a Tour de France team recently. His research and experience suggests that a ‘zone 2’ training program can restore mitochondrial function. Essentially, 4-5 times a week for 1-1.5 hrs, exercise with a heart rate in your zone 2.
Check out the work by Iñigo San Millán, Ph.D for more detail.
I found your article very informative I have fibromyalgia file 40 years .My son just had Covid again and he is exhibiting similar symptoms !Is there any solutions to this condition?
I am so troubled by this problem
I think that the hammer/nail analogy is apt. But my clinical experience also points to the “disease/defense” war analogy. Each “disease” (lack of ease) is evidence of a war between stress (infection/trauma/prolonged mental worry etc). I also believe that Selye,s discovery of an energy requiremenf to adapt to the stress (The General Adaptation Syndrome) is the key to ANY disease. This also makes the hammer/nail concept sensible. My guess (from personal research and clinical experience) is that the various forms of thiamine could be the “hammer”. The “illness” is evidence of a defense in process and should be assisted. If energy is the key, thiamine sits at the essential gate to energy synthesis. Anybody whose interest lies in this direction should surf ALL the posts on Hormones Matter. The details are all there and our book “Lonsdale D, Marrs C. Thiamine deficiency disease, dysautonomia and high calorie malnutrition” says it all!!
I have systemic parasites and believe they have been in my body since I was a teenager (I am nearly 60). I lost my energy around age 14 and have been dragging myself around ever since. My poor Mitochondria must be overwhelmed in a constant, unrelenting battle. The damn worms seem indestructible, like the monster that never dies in a horror movie. Anything I try to do to get rid of them just sends them into breeding and hatching overdrive and due to their unrelenting movement, if I get 3 hours sleep per night, it’s a miracle…….
Enlightening words that led me to seek information about the effect of low-level laser therapy–which I use (with incomplete understanding of its mechanism) to mediate muscle pain in my horses, hip dysplasia in my cat, and various mishaps in my own body–on mitochondria. One of the main mechanisms of low-level laser action occurs in the mitochondria, a fact that helps me understand why the therapy works so well. Thank you for your article.
By what mechanism does the laser therapy work?
A very interesting read. Fibromyalgia would certainly fall into this quite comfortably!
It would be wonderful to get your ‘take’ on this. One feels so helpless and it is extremely difficult for others to relate … as there aren’t ‘visible’ signs other than difficulty in movement etc, flare-ups!
Fibromyalgia most definitely fits into this category. I think it is one of the classic examples of mitochondrial damage or dysfunction even though it is not recognized as such.
Very interesting and informative article…greatly enjoyed reading it. Please can you advise me what supplements to take in order to help repair mitochondrial injury?
Thank you
Technically, I cannot as I am a PhD, not an MD, but if you read through our other articles on the topic, you’ll see that thiamine is critical, as are many of the B vitamins. Magnesium, CoQ10, L-carnitine, alpha lipoic acid and others are also important. It should be noted as well that vitamin D is often deficient, almost as a rule in most folks, but especially those who have chronic illness and who have been on medications chronically.
I found your article fascinating. I don’t have a science background beyond personal interest and journalistic research but with many decades of interest in health and body/mind, I feel increasingly that understanding physiological function metaphorically is going to be instrumental to understanding it properly, materially.
I have written some thoughts on possibly why cells become ‘rogue’ which resonates somewhat with your views on mitochondria and might be of interest to you:
http://rosross.wordpress.com/2014/02/26/you-and-your-body-are-in-this-together/
What an interesting perspective, especially the concept of memory. Indeed, cells have memory, multiple generations of memory, embedded. Many of the medications and environmental toxins silence or change those memories, and perhaps, that is where the problems emerge. Thank you for reading.
I just read “You and Your Body Are in This Together”, and what you say really resonates with me. Thank you. I agree, especially with your last two paragraphs.
I have come to a similar conclusion… after I began to dig deeper into everything my daughter was reacting to (salicylates in foods, pesticides, dry-cleaning chemicals, tylenol, vaccines)… I found a common thread- they are all Mitochondrial Toxins…
if the environment poisons our detector for environmental harm… is it possible for the cell danger response to NEVER turn off… I think it may be…